A subscription to JoVE is required to view this content. Sign in or start your free trial.
Integrated Photoacoustic, Ultrasound, and Angiographic Tomography (PAUSAT) for NonInvasive Whole-Brain Imaging of Ischemic Stroke
In This Article
Summary
This work demonstrates the use of a multimodal ultrasound-based imaging platform for noninvasive imaging of ischemic stroke. This system allows for the quantification of blood oxygenation through photoacoustic imaging and impaired perfusion in the brain through acoustic angiography.
Abstract
Presented here is an experimental ischemic stroke study using our newly developed noninvasive imaging system that integrates three acoustic-based imaging technologies: photoacoustic, ultrasound, and angiographic tomography (PAUSAT). Combining these three modalities helps acquire multi-spectral photoacoustic tomography (PAT) of the brain blood oxygenation, high-frequency ultrasound imaging of the brain tissue, and acoustic angiography of the cerebral blood perfusion. The multi-modal imaging platform allows the study of cerebral perfusion and oxygenation changes in the whole mouse brain after stroke. Two commonly used ischemic stroke models were evaluated: the permanent middle cerebral artery occlusion (pMCAO) model and the photothrombotic (PT) model. PAUSAT was used to image the same mouse brains before and after a stroke and quantitatively analyze both stroke models. This imaging system was able to clearly show the brain vascular changes after ischemic stroke, including significantly reduced blood perfusion and oxygenation in the stroke infarct region (ipsilateral) compared to the uninjured tissue (contralateral). The results were confirmed by both laser speckle contrast imaging and triphenyltetrazolium chloride (TTC) staining. Furthermore, stroke infarct volume in both stroke models was measured and validated by TTC staining as the ground truth. Through this study, we have demonstrated that PAUSAT can be a powerful tool in noninvasive and longitudinal preclinical studies of ischemic stroke.
Introduction
Blood transports oxygen (via the hemoglobin protein) and other important nutrients to tissues in our bodies. When the flow of blood through tissues is interrupted (ischemia), severe damage to the tissues can occur, the most immediate effects of which are due to a lack of oxygen (hypoxia). Ischemic stroke is the result of interrupted blood flow to a certain region of the brain. The brain damage resulting from an ischemic stroke can occur within minutes of a vessel blockage, and can often have debilitating and lasting effects1,2. A highly valuable strategy to evaluate the physiopathology after ischemic stroke and identify and test new treatments is the use of small-animal models in the lab. Treatments discovered in the lab aim to be translated to clinical use and improve patients' lives. However, the use of animals in biomedical research needs to be carefully evaluated according to Russell and Burch’s 3Rs principles: replacement, reduction, and refinement3. The objective of the reduction component is to reduce the number of animals without compromising data collection. With this in mind, being able to longitudinally evaluate the lesion evolution via noninvasive imaging allows a great advantage in reducing the number of animals required, as well as maximizing the information obtained from each animal4.
Photoacoustic tomography (PAT) is a hybrid imaging modality that combines optical absorption contrast with ultrasound imaging spatial resolution5. The imaging mechanism of PAT is as follows. An excitation laser pulse is illuminated on the target being imaged. Assuming the target absorbs light at the wavelength of the excitation laser, it will increase in temperature. This quick increase in temperature results in a thermoelastic expansion of the target. The expansion causes an ultrasound wave to propagate out from the target. By detecting the ultrasound wave at many positions, the time required for the wave to propagate from the target to the detectors can be used to create an image through a reconstruction algorithm. The ability of PAT to detect optical absorption in deep tissue regions differentiates PAT from ultrasound imaging, which detects boundaries of differing acoustic impedances of tissues5. In the visible and near-infrared spectra, the primary highly absorbing biomolecules that are abundant in organisms are hemoglobin, lipids, melanin, and water7. Of particular interest in the study of stroke is hemoglobin. Since oxyhemoglobin and deoxyhemoglobin have different optical absorption spectra, PAT can be used with multiple excitation laser wavelengths to determine the relative concentration of the two states of the protein. This allows the oxygen saturation of hemoglobin (sO2), or blood oxygenation, to be quantified in and outside of the infarct region8,9. This is an important measure in ischemic stroke, as it can indicate the level of oxygen in the damaged brain tissue following ischemia.
Acoustic angiography (AA) is a contrast-enhanced ultrasound imaging method that is particularly useful for imaging the morphology of vasculature in vivo10. The method relies on the use of a dual-element wobbler transducer (a low frequency element and a high frequency element) in conjunction with microbubbles injected into the circulatory system of the imaging subject. The low-frequency element of the transducer is used for transmitting at the resonant frequency of the microbubbles (e.g., 2 MHz), while the high-frequency element is used to receive the super harmonic signals of the microbubbles (e.g., 26 MHz). When excited at a resonant frequency, the microbubbles have a strong nonlinear response, resulting in the production of super harmonic signals that surrounding body tissues do not produce11. By receiving with a high-frequency element, this ensures that only the microbubble signals are detected. Since the microbubbles are confined to the blood vessels, the result is an angiographic image of blood vessel morphology. AA is a powerful method for imaging ischemic stroke, as the microbubbles that flow through the circulatory system are not be able to flow through blocked vessels. This allows AA to detect regions of the brain that are not perfused due to ischemic stroke, which indicates the infarct region.
Preclinical ischemic stroke research generally relies on the use of histology and behavioral testing to assess the location and severity of the stroke. Triphenyltetrazolium chloride (TTC) staining is a common histological analysis used to determine the stroke infarct volume. However, it can only be used at an endpoint, since it requires the animal to be euthanized12. Behavioral tests can be used to determine motor function impairment at multiple time points, but they cannot provide quantitative anatomical or physiological values13. Biomedical imaging provides a more quantitative approach to studying the effects of ischemic stroke noninvasively and longitudinally9,14,15. However, existing imaging technologies (such as small-animal magnetic resonance imaging [MRI]) can come at a high cost, be unable to provide concurrent structural and functional information, or have limited penetration depth (as most optical imaging techniques).
Here, we combine photoacoustic, ultrasound, and angiographic tomography (PAUSAT; see system diagram in Figure 1), which allows for complementary structural and functional information of blood perfusion and oxygenation following ischemic stroke16. These are two important aspects in assessing the severity of injury and monitoring the recovery or response to treatments. Using these integrated imaging methods can increase the amount of information obtained by each animal, reducing the number of animals required and providing more information in the study of potential treatments for ischemic stroke.
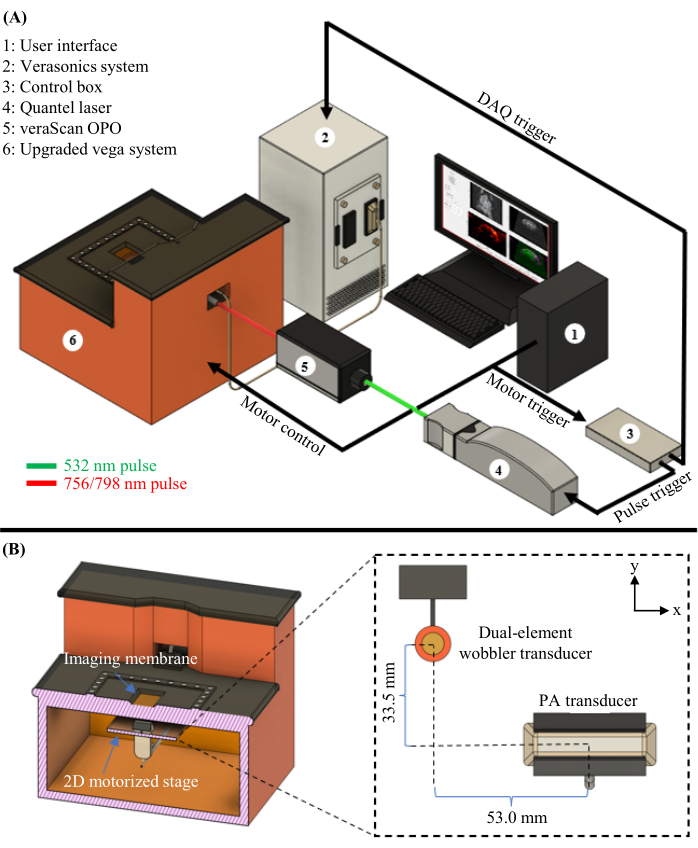
Figure 1: PAUSAT diagram. (A) Complete schematic of the PAUSAT system, including the laser and OPO used for PAT. (B) Inside view of the PAUSAT system, including two ultrasound transducers. The dual-element wobbler transducer is used for both B-mode ultrasound and AA, and the linear-array transducer is used for PAT. Both transducers are mounted on the same 2D motorized stage, allowing for scanning to generate volumetric data. This figure has been modified from16. Please click here to view a larger version of this figure.
Protocol
All animal procedures were approved by the Duke University Medical Center Animal Care and Use Committee and were conducted in accordance with the United States Public Health Service's Policy on Humane Care and Use of Laboratory Animals. Male and female C57BL/6J mice (see Table of Materials) were used for these studies. A minimum of three animals were imaged per stroke model group. See Figure 2 for the workflow followed in this protocol.
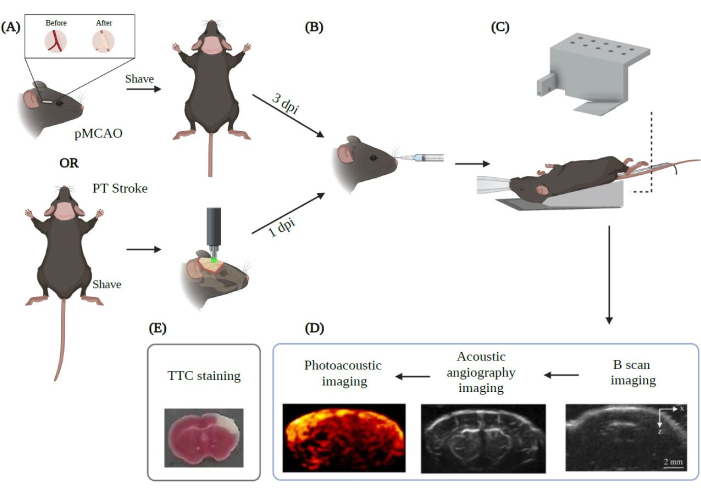
Figure 2: Summary of the experimental procedure for PAUSAT imaging applied to stroke. Created with Biorender.com. The figure shows the workflow of the imaging procedure starting from (A) the two main stroke models (pMCAO and PT stroke). (B) A retro-orbital injection of the microbubbles must be performed prior to positioning the animal on the PAUSAT membrane. (C) A mask providing continuous anesthesia and a heating pad to keep the animal's body temperature stable are required in this setup. The animal's body is placed on the heating pad while the head rests on the membrane of the system. (D) The order of image acquisition is also displayed in the figure. (E) TTC staining is performed to validate our results in this study. DPI: days post-injury. Please click here to view a larger version of this figure.
1. Inducing the stroke mouse model
- Permanent middle cerebral artery occlusion (pMCAO) with common carotid artery (CCA) ligation.
NOTE: Briefly, perform permanent ligation of the right CCA and posterior electrocauterization of the right middle cerebral artery (MCA)17. This procedure limits the cerebral blood flow in the right cortex of the brain, causing an ischemic stroke18.- Induce anesthesia in an induction chamber using an inhalational mix of 5.0% isoflurane in 30% O2/70% N2 until loss of consciousness (recognized as a loss of the pedal reflex).
- Intubate the animal using a 20 G catheter (Table of Materials) and connect it to an automatic ventilator. Set the flow rate based on the animal's body weight and keep the animal anesthetized using 1.5%-2.0% isoflurane in 30% O2/70% N2.
- Using a heating lamp and a rectal probe connected to a temperature controller device, keep the animal's body temperature at 37 °C.
- Put a drop of lubricant eye ointment on the mouse's eyes.
- Place the animal in a supine position and remove the hair from the neck area using a hair trimmer.
- Clean the skin area by first using a cotton swab with povidone-iodine, and then using a sterile pad with 70% ethanol. Perform this thrice.
- Verify the depth of anesthesia and the absence of pain by slightly pinching the animal's posterior paw.
- Make a 0.8 cm sagittal incision on the middle line of the neck and expose the right CCA.
- Prepare a suture for the CCA ligation by dissociating a 4-0 silk suture into thinner threads that compose the main thread. Use a length of 1.5 cm of one of the subthreads to permanently ligate the CCA.
NOTE: After tightening the knot, remove excess thread by cutting the extension at a 1-2 mm distance to the knot. - Apply a drop of bupivacaine before closing the wound.
- Close the incision using interrupted 4-0 silk surgical sutures and apply triple antibiotic ointment on the surface to prevent infection.
- Move the mouse to expose the right lateral side of the animal's body.
-
Remove the hair in the region between the ear and the eye area using a hair trimmer.
-
Disinfect the surgical area using a cotton swab with povidone-iodine, followed by a sterile pad with 70% ethanol. Repeat this step three times.
-
Place a sterile drape to secure the surgical area. Then, make a 0.5 cm incision between the right eye and ear of the animal, exposing the joint between the skull and the temporal muscle.
- Using a cautery loop, cauterize the muscle to separate it from the skull and expose the area of the MCA.
- Drill a 0.2 mm2 window to expose the MCA using an electric drill, and use electrocautery on the MCA to occlude blood flow.
NOTE: A single pulse at 80% power intensity is enough to cauterize the MCA. - Using a 1mL syringe attached to a 27G needle, apply a drop of bupivacaine (Table of Materials) at the surgical site.
- Close the skin incision using interrupted 6-0 clear monofilament sutures and apply triple antibiotic ointment on the surface to prevent infection.
- After completing the surgery, transfer the animal to an incubator with controlled temperature (32 °C) and allow the animal to recover.
- After 2 h, transfer the animal to its home cage and provide food and water ad libitum.
- Photothrombotic stroke (PT stroke)
NOTE: Briefly, PT stroke is performed by illuminating Rose Bengal within the vessels in the brain. Rose Bengal is administered intraperitoneally, and once it has been well distributed throughout the body (5 min), it is illuminated by a green cold light, which activates the Rose Bengal to generate reactive oxygen species (ROS). These ROS damage the membrane of endothelial cells, creating thrombi within the entire illuminated area and leading to local cerebral blood flow disruption19.- Induce anesthesia in an induction chamber using an inhalational mix of 5.0% isoflurane in 30% O2/70% N2 until loss of consciousness (recognized as a loss of the pedal reflex).
- Set the animal to a stereotaxic frame, keeping the animal anesthetized using a mask and 1.5%-2.0% isoflurane in 30% O2/70% N2.
- Keep the animal at 37 °C using a hot water recirculating heater and a rectal probe to measure the animal's body temperature.
- Put a drop of lubricant eye ointment on the mouse's eyes.
- Shave the animal's head using a hair trimmer.
- Clean the shaved scalp area three times, first using a cotton swab with povidone-iodine, and then using a sterile pad with 70% ethanol.
- Verify the absence of pain by slightly pinching the animal's posterior paw.
- Make a 1.4 cm sagittal incision on the middle line of the scalp using a scalpel and expose the skull.
- Using a sharp pencil, make a mark at 1.5 mm from the bregma toward the right side.
- Place a circular pinhole of 2.5 mm diameter centered on the 1.5 mm mark.
NOTE: A square containing a circular pinhole can be made by using double-sided black tape and making a 2.5 mm diameter opening in the center by using a single-hole punch tool of the mentioned size. - Place the green cold light on the circular pinhole, keeping the gap between the light and the pinhole to a minimum.
- Cover the area using aluminum foil to avoid spreading of the light.
- Once the setup is ready, inject the animal intraperitoneally with 10 mg/kg Rose Bengal (10 mg/mL in 1x phosphate-buffered saline [PBS]) and wait for 5 min.
- After 5 min, turn on the cold light source (intensity: 4.25) and maintain the exposure for 15 min.
- Next, turn off the cold light and verify the stroke either by the naked eye (the area is expected to be whiter than the surrounding area) or using external devices to measure the cerebral blood flow (for example, by using laser speckle contrast imaging (Table of Materials; see step 5.1).
- Using a 1mL syringe attached to a 27G needle, apply a drop of bupivacaine (Table of Materials) at the surgical site.
- Close the skin incision using interrupted 6-0 clear monofilament sutures, and apply triple antibiotic ointment on the surface to prevent infection.
- After completing the surgery, transfer the animal to an incubator with controlled temperature (32 °C) and allow the animal to recover.
- After 2 h, transfer the animal to its home cage and provide food and water ad libitum.
2. Preparing PAUSAT for imaging
- Turn on the 532 nm laser and leave it on for 15 min to warm-up.
- Prepare the imaging platform for the anesthetized animal.
- Place a customized ramp (Figure 2C) attached to the manually adjustable stage (Table of Materials) beside the imaging membrane.
- Attach a mouse tooth-holder with the breathing tube connected to the customized ramp and secure a heating pad on the surface of the ramp.
- After the laser has warmed up, check that the laser path and coupling into the fiber-bundle is well-aligned using a near-infrared detector card (Table of Materials) by placing the card in front of the fiber-bundle input and ensuring the laser light enters the bundle.
NOTE: Adjust any laser path mirrors as necessary to ensure the laser input is centered with the fiber-bundle input.
3. Preparing the animal for PAUSAT
NOTE: PAUSAT is performed 1 day following PT stroke surgery or 3 days following pMCAO surgery. Preparing PAUSAT for imaging (step 2) takes about 20 min and should be done immediately before preparing the animal for PAUSAT.
- Induce anesthesia in an induction chamber using an inhalation mix of 5% isoflurane mixed with 30% O2/70% N2 until loss of consciousness (recognized as a loss of the pedal reflex).
- Transfer the animal to a heated platform with a tooth-holder and a mask, and maintain the anesthesia at 1.5%-2.0% isoflurane in 30% O2/70% N2.
- Use a heating lamp and a rectal probe connected to a temperature controller device to keep the animal's body temperature at 37 °C.
- Trim the hair on the top of the animal's head by using an electric shaver. Include the region from near the eyes to behind the ears.
- Shave the hair on the top of the animal's head by applying a commercial hair removal cream to completely remove the remaining short hair. Leave on the skin for 5-6 min, then wipe off using a cotton swab dabbed in water to help fully remove the cream. Repeat until the skin is clear of hair.
NOTE: For imaging 1 day after the surgery, these steps can be performed before starting the surgery; at 1 day after PT stroke, they can be omitted. When PAUSAT image acquisition is performed several days after the surgery, this step is critically required to be performed. - Once the animal and the system are ready for imaging, and right before transferring the animal to the system's platform, inject a 100 µL solution of microbubbles at the stock concentration (Table of Materials) retro-orbitally using a 27 G needle.
NOTE: Once the bubbles are in circulation in the bloodstream, there is a limited amount of time to image without a significant loss of signal (~10 min). - Put a drop of eye protection lotion on the mouse's eyes.
NOTE: It is not recommended to apply eye lubricant until the retro-orbital injection is performed to avoid foreign substances reaching the animal's bloodstream. Therefore, hair removal cream application must be performed slowly and carefully to avoid getting too close to the eyes (but enough to expose the region of interest where the stroke is expected). The hair cream removal is performed with a cotton swab previously dabbed in water, preventing the cream from dripping, which can damage the eyes.
4. PAUSAT Imaging
NOTE: This is done to image the contra- and ipsi-lateral regions of the brain after stroke
- Transfer the mouse to the integrated PAUSAT (Table of Materials) image platform, placing the mouse in a supine position on the customized ramp (Figure 2C).
- Fill the imaging window with enough distilled water on the surface for acoustic coupling.
NOTE: An optional ramp-printed using a 3D printer-is recommended to prevent the animal's body from getting wet during the image acquisition and improve the animal's comfort. It also helps maintain a stable body temperature. Furthermore, the ramp can be attached to a manual stage (Table of Materials) to adjust the focal depth of the dual-element wobbler transducer relative to the mouse head. The custom ramp design file is available upon request to the authors. - Secure the mouse head in the tooth-holder and ensure proper anesthesia and air flow.
- Using a heating lamp and a rectal probe connected to a temperature controller device, keep the animal's body temperature at 37 °C.
- Open the imaging application (Table of Materials) and navigate to B-mode ultrasound.
- Use the live ultrasound window to manually adjust the mouse head to the desired position.
- Use the live ultrasound window to adjust the height of the stage, such that the focal depth of the transducer (19 mm) is approximately in the middle of the area to be imaged.
- Imaging with B-mode ultrasound
- Adjust the value of the ultrasound transmission frequency in B-mode (for these studies, use 16 MHz).
- Input the save directory information in the imaging application.
- Use the floating box to select the desired region for the B-mode scan of the brain.
- Press the Acquire Static button.
- Check the results of the scan in the application once the image acquisition is completed to ensure that the desired region has been imaged.
NOTE: Avoid unnecessary delays in B-mode imaging acquisition to ensure that a high enough concentration of microbubbles remains in the bloodstream for the AA.
- Imaging with AA
- Return to Image Acquisition.
- Change to Acoustic Angiography mode in the imaging application (Table of Materials).
- Input the desired scan protocol parameters (the most important of which is frame spacing and the number of frames per position, which was set to 0.2 mm and 10, respectively, for these studies).
- Press the Acquire Static button.
NOTE: The AA acquisition takes longer than the B-mode ultrasound. - Once the scan is completed, check the results of the scan under Image Analysis to ensure that the image quality is as expected.
NOTE: For AA mode, a more representative whole-brain volume can be acquired by repeating a second scan at a different focal depth inside the brain and later recombining the images with proper post-processing (see Figure 3).
- Imaging with photoacoustic tomography
- Open the optical parametric oscillator (OPO) application (Table of Materials) and set it to 756 nm.
NOTE: OPOs can easily get out of calibration, so prior to the experiment, ensure that the OPO is calibrated correctly by using an independent spectrometer. - Manually translate the linear-array transducer to the previously determined coordinates to ensure that the wobbler volumes and the linear-array volumes are automatically co-registered.
NOTE: It is critical that a co-registration experiment using a phantom grid is done beforehand to determine the exact distance needed to translate the stage, such that the resulting data from both transducers are co-registered in 3D. - Open the laser application and turn on the 532 nm laser.
- Using a laser power meter, measure the energy of the laser output and ensure it is the desired energy (~10 mJ per pulse was used for these studies).
- Select the desired scanning parameters for PAT (0.4 mm step size, 20 mm scan length, and 10 frames averaged per position).
- Open the ultrasound data acquisition system MATLAB program (Table of Materials) and press the Run button.
- Acquire the PAT scan by pressing the Start button.
- Once the scan is complete, open the MATLAB save program. Change the save name to the desired file name and press the Run button.
- Change the OPO wavelength to 798 nm and repeat the steps from 4.10.3 to 4.10.8.
NOTE: For a longitudinal study, it is recommended to allow the animal to recover by placing it in an incubator and under observation for a few hours (following steps 1.1.18 and 1.1.19). If result validation is desired, continue to section 5 immediately after PAUSAT imaging.
- Open the optical parametric oscillator (OPO) application (Table of Materials) and set it to 756 nm.
5. Optional: Results validation
- Laser speckle contrast imaging (LSCI).
- Anesthetize the animal using 1.5%-2.0% isoflurane in 30% O2/70% N2.
- Set the animal to a stereotaxic frame, keeping the animal anesthetized using a mask and the above-mentioned inhalational anesthesia.
- Keep the animal at 37 °C using a hot water recirculating heater and a rectal probe to measure the animal's body temperature.
- Put a drop of eye protection lotion on the mouse's eyes.
- Verify the absence of pain by slightly pinching the animal's posterior paw.
- Remove the hair on the animal's scalp using a hair trimmer.
- Disinfect the surgical area using a cotton swab with povidone-iodine, followed by a sterile pad with 70% ethanol. Repeat this step three times.
- Make a 1.4 mm sagittal incision on the middle line of the scalp and expose the skull. Use forceps to hold the scalp and prevent it from occupying the area of the brain to scan.
- Apply some drops of saline on the skull and place the laser speckle contrast system device (Table of Materials) over the animal's head.
- Under the File menu, set the device in Online mode, contained in the submenu Working Mode.
- Select the default image storage folder in the File menu and the Save Settings submenu.
- In the Light Source menu, connect the guiding laser ("Laser on") and the white light ("White-light on") to locate the imaging window at the right position.
- In the Setting menu, select Magnification settings, move the cursor manually to 2.5, and press Apply and OK to save the settings.
- Adjust the focus by manually moving the focus bar located in the top submenu of the main page.
- In the Setting menu, select Pseudo Color Threshold Setting, adjust the threshold as desired, and press Apply and OK to save the settings.
- In the Light Source menu, disconnect the guiding laser ("Laser off") and the white light ("White-light off") before capturing the image.
- Capture the image by selecting the Play symbol in the top submenu of the main page.
- Triphenyltetrazolium chloride (TTC) staining
- Deeply anesthetize the animal using 5% isoflurane in 30% O2/70% N2.
- Once the animal has stopped breathing, decapitate it using sharp scissors.
- Remove all the skin around the head and the muscles in the neck area.
- Make a sagittal cut in the occipital part of the skull until it reaches the parietal bone.
- Make a horizontal cut (~5 mm) in the left and right side below the blood vessel. Remove the occipital bone of the skull using straight forceps.
- Make a cut (~5 mm) at the frontonasal suture of the skull.
- Make a sagittal cut (~10-15 mm) in the midline of the skull-between hemispheres-and make sure they are completely separated.
- Using size #7 curved scissors, remove the parietal left and right bones of the skull from the center to the sides.
- Transfer the brain to a container filled with 5 mL of ice-cold 1x PBS and keep it on ice for 10 min.
- Transfer the brain to a stainless-steel brain matrix (1 mm thick sections).
- Section the brain in 1 mm coronal sections using disposable razor blades (Table of Materials).
- Holding the blades by their sides, transfer into a container filled with ice-cold 1x PBS.
- Carefully separate the sections from the blades one by one.
- Transfer the brain slices into a 70 mm diameter Petri dish containing 5 mL of 2% TTC (Table of Materials, 3) in 1x PBS.
- Incubate for 15 min in the dark at room temperature (R/T).
- After 15 min, discard the TTC, replace it with 3 mL of formalin, and incubate in the dark for at least 30 min at R/T.
- Finally, transfer the brain slices onto a transparent plastic film and scan the samples, including a ruler in the scan image as a reference for future measurements.
Results
Imaging of blood vessel morphology in the brain
AA generates blood vessel morphology images by exciting microbubbles in the circulatory system at their resonant frequency and receiving the super harmonic response of the microbubbles. By using the customized ramp (Figure 2C) attached to a manually adjustable stage, we can image the mouse brain with AA mode at two different focal depths. When deeper regions are targeted, more superficial regions (such as the cerebral cor...
Discussion
There are a few vital aspects of this method that, if done incorrectly, can lead to significantly decreased image quality and quantitative analysis. The most commonly occurring result of user-error in PAUSAT images is either a lack of signal or very low signal strength, both of which can occur for a variety of reasons. One such reason is a problem with the acoustic coupling. Large air bubbles in the water surrounding the mouse's head during imaging can often block the ultrasound from travelling to or from the transdu...
Disclosures
The authors declare no conflict of interest in this work.
Acknowledgements
The authors would like to acknowledge the engineering team at SonoVol Inc. for their technical support. This work was partially sponsored by the American Heart Association Collaborative Sciences Award (18CSA34080277), to J. Yao and W. Yang; The United States National Institutes of Health (NIH) grants R21EB027981, R21 EB027304, RF1 NS115581 (BRAIN Initiative), R01 NS111039, R01 EB028143; The United States National Science Foundation (NSF) CAREER award 2144788; the Chan Zuckerberg Initiative Grant (2020-226178), to J. Yao; and NIH grants R21NS127163 and R01NS099590 to W. Yang.
Materials
| Name | Company | Catalog Number | Comments |
| 20 GA catheter | BD Insyte Autoguard Winged | 381534 | For mouse intubation |
| 2,3,5-Triphenyltetrazolium chloride | Sigma | T8877 | Necessary for TTC-staining brain for validation |
| 532nm Laser | Quantel | Q-smart 850 | Laser used to pump the OPO for PAT |
| Automatic Ventilator Rovent Jr. | Kent Scientific | RV-JR | To keep mice under anesthesia during surgical procedure |
| Black braided silk 4-0 USP | Surgical Specialties | SP116 | Used for sutures on the neck for pMCAO surgery |
| Bupivacaine | Hospira | 0409-1159-18 | Used prior to closing wounds during surgical procedure |
| C57BL/6 Mice | Jackson Lab | #000664 | Mice used for studying ischemic stroke (2-6 month old male/female) |
| Clear suture | Ethicon | 8606 | Used for closing wound (PT stroke and pMCAO). A clear suture won't interfere with PAT |
| Cold Light LED | Schott | KL 1600 | Needed to create PT stroke |
| Disposable Razor Blade | Accutec Blades | 74-0002 | For sectioning mouse brain |
| Electric drill | JSDA | JD-700 | Used to expose MCA during pMCAO procedure |
| Electrocauterization tool | Wet-Field | Wet-Field Bipolar-RG | Stops blood flow after drilling during pMCAO procedure |
| Hair removal gel | Veet | 8282651 | Used to remove hair from mouse prior to imaging |
| High Temperature Cautery Loop Tip | BOVIE Medical Corporation | REF AA03 | Used to avoid bleeding when separating the temporal muscle from the skull |
| IR Detector Card | Thorlabs | VRC5 | Used to ensure light path is aligned |
| Laser Power Meter | Ophir | StarBright, P/N 7Z01580 | Can be used to calibrate the laser energy prior to imaging |
| Laser Speckle Imaging System | RWD Life Science Co. | RFLSI-III | Can be used to validate stroke surgery success |
| Lubricant Eye Ointment | Soothe | AB31336 | Can be used to avoid drying of the eyes |
| Manually adjustable stage | Thorlabs | L490 | Used with custom ramp for multiple focal depth AA imaging |
| Modified Vega Imaging System | Perkin Elmer | LLA00061 | System containing both B-mode/AA and PAT transducers |
| Optical Parametric Oscillator | Quantel | versaScan-L532 | Allows for tuning of excitation wavelength in a large range |
| Programmable Ultrasound System | Verasonics | Vantage 256 | Used for PAT part of system |
| Rose Bengal | Sigma | 330000 | Necessary to induce PT stroke |
| Suture | LOOK | SP116 | Used for permanent ligation of CCA |
| Temperature Contoller | Physitemp | TCAT-2 | Used to maintain stable body temperature of mice during procedures |
| VesselVue Microbubbles | Perkin Elmer | P-4007001 | Used for acoustic angiography (2.43 × 10^9 microbubbles/mL) |
References
- Durukan, A., Tatlisumak, T. Acute ischemic stroke: overview of major experimental rodent models, pathophysiology, and therapy of focal cerebral ischemia. Pharmacology Biochemistry and Behavior. 87 (1), 179-197 (2007).
- Vander Worp, H. B., van Gijn, J. Clinical Practice. Acute ischemic stroke. The New England Journal of Medicine. 357 (6), 572-579 (2007).
- Tannenbaum, J., Bennett, B. T. Russell and Burch's 3Rs then and now: the need for clarity in definition and purpose. Journal of the American Association for Laboratory Animal Science. 54 (2), 120-132 (2015).
- Hochrainer, K., Yang, W. Stroke proteomics: from discovery to diagnostic and therapeutic applications. Circulation Research. 130 (8), 1145-1166 (2022).
- Wang, L. V., Yao, J. A practical guide to photoacoustic tomography in the life sciences. Nature Methods. 13 (8), 627-638 (2016).
- Aldrich, J. E. Basic physics of ultrasound imaging. Critical Care Medicine. 35 (5), S131-S137 (2007).
- Jacques, S. L. Optical properties of biological tissues: a review. Physics in Medicine and Biology. 58 (11), R37-R61 (2013).
- Li, M., Tang, Y., Yao, J. Photoacoustic tomography of blood oxygenation: a mini review. Photoacoustics. 10, 65-73 (2018).
- Menozzi, L., Yang, W., Feng, W., Yao, J. Sound out the impaired perfusion: Photoacoustic imaging in preclinical ischemic stroke. Frontiers in Neuroscience. 16, 1055552 (2022).
- Gessner, R. C., Frederick, C. B., Foster, F. S., Dayton, P. A. Acoustic angiography: a new imaging modality for assessing microvasculature architecture. International Journal of Biomedical Imaging. 2013, 936593 (2013).
- Dayton, P. A., Rychak, J. J. Molecular ultrasound imaging using microbubble contrast agents. Frontiers in Bioscience. 12, 5124-5142 (2007).
- Isayama, K., Pitts, L. H., Nishimura, M. C. Evaluation of 2, 3, 5-triphenyitetrazolium chloride staining to delineate rat brain infarcts. Stroke. 22 (11), 1394-1398 (1991).
- Ruan, J., Yao, Y. Behavioral tests in rodent models of stroke. Brain Hemorrhages. 1 (4), 171-184 (2020).
- Parthasarathy, A. B., Kazmi, S. M. S., Dunn, A. K. Quantitative imaging of ischemic stroke through thinned skull in mice with Multi Exposure Speckle Imaging. Biomedical Optics Express. 1 (1), 246-259 (2010).
- Hingot, V., et al. Early ultrafast ultrasound imaging of cerebral perfusion correlates with ischemic stroke outcomes and responses to treatment in mice. Theranostics. 10 (17), 7480-7491 (2020).
- Menozzi, L., et al. Three-dimensional non-invasive brain imaging of ischemic stroke by integrated photoacoustic, ultrasound and angiographic tomography (PAUSAT). Photoacoustics. 29, 100444 (2022).
- Llovera, G., Roth, S., Plesnila, N., Veltkamp, R., Liesz, A. Modeling stroke in mice: permanent coagulation of the distal middle cerebral artery. Journal of Visualized Experiments. (89), e51729 (2014).
- Trotman-Lucas, M., Kelly, M. E., Janus, J., Fern, R., Gibson, C. L. An alternative surgical approach reduces variability following filament induction of experimental stroke in mice. Disease Models & Mechanisms. 10 (7), 931-938 (2017).
- Labat-Gest, V., Tomasi, S. Photothrombotic ischemia: a minimally invasive and reproducible photochemical cortical lesion model for mouse stroke studies. Journal of Visualized Experiments. (76), e50370 (2013).
- Matsumoto, Y., et al. Visualising peripheral arterioles and venules through high-resolution and large-area photoacoustic imaging. Scientific Reports. 8 (1), 14930 (2018).
- Xu, Y., Wang, L. V., Ambartsoumian, G., Kuchment, P. Reconstructions in limited-view thermoacoustic tomography. Medical Physics. 31 (4), 724-733 (2004).
- Yal Tang, ., et al. High-fidelity deep functional photoacoustic tomography enhanced by virtual point sources. Photoacoustics. 29, 100450 (2023).
- Zheng, W., Huang, C., Zhang, H., Xia, J. Slit-based photoacoustic tomography with co-planar light illumination and acoustic detection for high-resolution vascular imaging in human using a linear transducer array. Biomedical Engineering Letters. 12 (2), 125-133 (2022).
- Wang, Y., et al. Slit-enabled linear-array photoacoustic tomography with near isotropic spatial resolution in three dimensions. Optics Letters. 41 (1), 127-130 (2016).
- Vu, T., Li, M., Humayun, H., Zhou, Y., Yao, J. A generative adversarial network for artifact removal in photoacoustic computed tomography with a linear-array transducer. Experimental Biology and Medicine. 245 (7), 597-605 (2020).
- Zhang, H., et al. Deep-E: A fully-dense neural network for improving the elevation resolution in linear-array-based photoacoustic tomography. IEEE Transactions on Medical Imaging. 41 (5), 1279-1288 (2022).
- Hauptmann, A., et al. Model-based learning for accelerated, limited-view 3-D photoacoustic tomography. IEEE Transactions on Medical Imaging. 37 (6), 1382-1393 (2018).
- Li, M., et al. Three-dimensional deep-tissue functional and molecular imaging by integrated photoacoustic, ultrasound, and angiographic tomography (PAUSAT). IEEE Transactions on Medical Imaging. 41 (10), 2704-2714 (2022).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved