A subscription to JoVE is required to view this content. Sign in or start your free trial.
Tuina Manipulation to Reduce Inflammation and Cartilage Loss in Knee Osteoarthritis Rats
In This Article
Summary
We present a protocol for Tuina manipulation performed on plaster-immobilized-induced knee osteoarthritis rats. Based on the preliminary results, it suggested that the efficacy of the method relied on the reduction of inflammation and cartilage loss.
Abstract
Clinical trials suggest that Tuina manipulation is effective in treating knee osteoarthritis (KOA), while further studies are required to discover its mechanism. Therefore, the manipulation of animal models of knee osteoarthritis is critical. This protocol provides a standard process for Tuina manipulation on KOA rats and a preliminary exploration of the mechanism of Tuina for KOA. The press and kneading manipulation method (a kind of Tuina manipulation that refers to pressing and kneading the specific area of the body surface) is applied on 5 acupoints around the knee joint of rats. The force and frequency of the manipulation were standardized by finger pressure recordings, and the position of the rat during manipulation is described in detail in the protocol. The effect of manipulation can be measured by pain behavior tests and microscopic findings in synovial and cartilage. KOA rats showed significant improvement in pain behavior. The synovial tissue inflammatory infiltration was reduced in the Tuina group, and the expression of tumor necrosis factor (TNF)-α was significantly lower. Compared to the control group, chondrocyte apoptosis was less in the Tuina group. This study provides a standardized protocol for Tuina manipulation on KOA rats and preliminary proof that the therapeutic effects of Tuina may be related to reducing synovial inflammation and delayed chondrocyte apoptosis.
Introduction
Knee osteoarthritis (KOA) is a degenerative disease mainly manifested in joint pain. Fibrosis, cracking, ulceration, and loss of articular cartilage are the main causes of this disease1. KOA has a high prevalence and may result in a profound impact on the daily life of patients, causing disability in severe cases. Among people aged 45-84 years, the prevalence of KOA increases with age, and the prevalence among people aged 85 years and above is 15%, with a predominance in women2,3. In addition, KOA might bring a serious economic burden on both the individual and the society. A survey showed that direct health care costs on KOA per capita reached up to $8,858 ± $5,120 per year4. With the aging of society, KOA has become a worldwide health problem and a major social issue, as well as a topical issue for scientific research.
Evidence-based studies have shown the effectiveness of Tuina manipulation in treating KOA5. Tuina manipulation could relieve pain and improve dysfunction in KOA patients, the mechanism of which is related to anti-inflammatory effects6,7. Scholars found that Tuina manipulation effectively inhibited the expression of inflammatory factors interleukin (IL)-β and 5-hydroxytryptamine and slowed down the degeneration of articular cartilage in a rabbit KOA model8. It suggested that Tuina could promote blood circulation and metabolism at the lesion site, which helped clear inflammatory factors such as IL-1, IL-6 and tumor necrosis factor (TNF)-α, thereby alleviating the clinical symptoms of KOA9. In addition, the passive movement of the joint through Tuina manipulation can promote the penetration and diffusion of synovial fluid into the articular cartilage and improves tissue nutrient metabolism10. Other studies suggested that Tuina manipulation can effectively improve the biomechanical indexes in KOA patients11. Manipulations applied on soft tissues can improve stress distribution over limbs and enhance balance function12,13. At the same time, with some joint adjustment manipulations, the alignment of the lower limbs can also be adjusted to correct abnormal gaits14,15.
The mechanism of action of Tuina manipulation in treating KOA remains to be explored, and therefore, an experimental study is necessary. The key to the application of Tuina in experimental animals is the standardization of modeling, animal fixation and intervention methods16. The modeling method determines whether the experimental animal can exhibit the characteristics of the disease. Meanwhile, appropriate fixation methods can facilitate the intervention of the Tuina manipulation and better reflect the effect of Tuina. The standardization of intervention methods is the most difficult part of Tuina manipulation. In 2010, the basic system of Chinese acupuncture standards mentioned the acupuncture point standards for experimental animals, providing the possibility for acupuncture and Tuina operations in animal experiments17. However, there are still difficulties in standardizing Tuina manipulation. There are multiple types of Tuina manipulation18. The choice of the specific manipulation mainly depends on the disease to be treated and the therapeutic theories the performer prefers. In the study of Tuina for KOA, more attention has been paid to the point-pressing manipulation (pressing the specific acupoints with the thumb or elbow), Yizhichan pushing manipulation (a pushing manipulation by wiggling the thumb), and press and kneading manipulation (which refers to pressing and kneading the specific area of the body surface by finger or palm)19. Press and kneading manipulation is one of the most widely used Tuina manipulations, which combines pressing and kneading to move the subcutaneous tissue20. Press and kneading manipulation applied on acupoints can promote blood circulation and relieve pain and represents the therapeutic effect of Tuina on KOA19.
In this protocol, the operation of press and kneading manipulation on KOA rats will be described in detail, including selected acupoints, intensity and frequency of the manipulation and the body position of the rat, so as to provide a reference for future research.
Protocol
This study has passed the animal ethics review conducted by the experimental animal ethics committee of Yueyang Hospital of integrated traditional Chinese and western medicine affiliated with Shanghai University of Traditional Chinese Medicine (YYLAC-2022-166).
1. Experimental animal preparation and grouping
- Animal Preparation
- Rear a total of 10 healthy SPF SD female rats of 200-220 g at room temperature (18-21 °C), humidity 40%-50%, 12 h: 12 h circadian rhythm alternations. Conduct pain-related animal experiments in strict compliance with the relevant provisions of animal ethical guidelines and guidelines.
- Grouping of animals
- Randomly divide the rats into the Tuina group and control group. Treat rats of the Tuina group with press and kneading manipulation for 21 days after modeling. Put rats of the control group in the same Tuina room and place them in a black cloth bag simultaneously while the Tuina group is undergoing treatment.
2. Modeling of animals
- Anesthetizing the animal
- Use isoflurane for gas anesthesia. Place the rat in the induction box with an induction concentration of 3%. Wobble the box after laying the rat down and confirm the anesthetization when the rat rollover with no attempt to return to the prone position.
- Remove the rat from the induction box and fix its nose in the anesthetic mask. Adjust the isoflurane concentration to 2% to maintain anesthesia. Confirm the anesthetization when the rat does not respond when pinching its paws. Apply eye ointment to the rats to prevent dryness when rats are anesthetized, as the eyelids cannot be closed.
- Modeling method21
- Use shaving machine to remove the hair of the right hind limb. Place a medical cotton pad between the right ankle and hip joint of the rat. Fix the right knee joint at 180° extension with 5-6 layers of wet plaster bandage evenly. Spiral wrap the plaster bandage starting from the ankle and covering 1/3 of the previous one. Use a hair dryer to dry and harden the plaster.
- External wrap the plaster with denture base material after the plaster bandage dries and hardens to fix the plaster and prevent it from gnawing.
- Mix the denture base materials to make it sticky and adhere the mixture to the outside of the plaster (the mixture should not exceed the edge of the bandage, Figure 1). After the mixture becomes hard, turn off the anesthesia machine and wait for the animal to wake up naturally. Supervise the rats to prevent anesthesia accidents before the rats wake up.
- Fix the plaster appropriately on the right hind limb of the rat, as a tight fixation restricts blood circulation, while a loose fixation tends to fall off. Observe the blood circulation in the terminal limb. If swelling of the terminal extremity or a purple complexion is detected, promptly cut off part of the plaster to help restore circulation. Remake the plaster if it is broken and fails to maintain the lower limb extension.
- Remove the plaster after 3 weeks of continuous immobilization. Use surgical scissors to cut off the denture base material outside and plaster bandage. Rinse the lower limb of the rat with saline and dry it with gauze. If there are local skin lesions, sterilize with iodophor.
- Model verification22
- X-ray-based verification
- Perform an X-ray examination of the right knee 1 day after the end of modeling. Take anteroposterior radiographs in the supine position with hip flexion at 30°, knee extension at 0°, and hip abduction at 15°. Keep the patella directly in front of the knee and put the radiator tube 110 mm from the knee joint.
- Take lateral radiographs in the right lateral decubitus position with right hip flexion at 30° and right knee extension at 0°. Make the left limb hip flexion at 70°, and knee flexion at 45°, and put the radiator tube 110 mm away from the knee joint. Set the detection parameters as exposure voltage 50 kV, current 250 mA, exposure dose 32 mAs, and exposure time 128 ms.
- Compare with normal rats' X-ray, check that the modeled knee X-ray shows narrower joint space with osteophyte hyperplasia at the edge.
- Osteoarthritis Research Society International (OARSI) scoring23
- Place the rat in the euthanasia box and perfuse CO2 at the rate of 30%-70% cage volume per minute. Stop perfusing CO2 after detecting that the rat is immobile, not breathing, and that the pupil is dilated. Observe for another 2 min to confirm death.
NOTE: Cervical dislocation can be performed after CO2-based euthanasia as a secondary form to confirm death. Fix the rat on the table and grasp its tail with one hand. Press down on the head of the rat with the thumb and index finger of the other hand. Confirm death when hearing the sound of a crack, and the rat loses movement and heartbeat at the same time. - Fix the rat in a supine position with a syringe needle on a foam board with the right hind limb flexed in abduction and external rotation. Pinch up the skin around the knee joint with surgical scissors. Expose the muscles around the knee joint by cutting the skin and then cutting the subcutaneous fascia.
- Cut off the femur and tibia diaphysis with bone scissors and remove the right knee joint. Gently remove the extra soft tissues, such as muscles and ligaments, outside the joint.
- Fix the joint in 4% paraformaldehyde for 24-48 h at 4 °C. Decalcify the joint in 10% formic acid solution for 3 days until the bone tissue can be easily poked with a needle.
- Place and trim the decalcified tissue in the fume hood and transfer it to a dehydration box in the dehydration machine. Add 75% ethanol for 4 h, then 90% ethanol for 2 h, followed by 95% ethanol for 1 h, absolute ethanol for 30 min, another round of fresh absolute ethanol for 30 min, alcohol benzene for 5-10 min, xylene for 5-10 min, another round of fresh xylene for 5-10 min, wax for 1 h, another round of fresh wax for 1 h, and final round of fresh wax for 1 h for dehydration and transparent wax immersion.
- Then, place the tissue into the machine for embedding. Cut the wax block into wax slices of 4 µm after the paraffin has set and flatten the slice in warm water. Place the slice on a glass slide and dry. Store it at room temperature.
- Observe the cartilage sample and score it according to the OA cartilage histopathology grade assessment (Table 1)23. If the score of rats after modeling is significantly higher than that of the normal rats, modeling was successful.
- Place the rat in the euthanasia box and perfuse CO2 at the rate of 30%-70% cage volume per minute. Stop perfusing CO2 after detecting that the rat is immobile, not breathing, and that the pupil is dilated. Observe for another 2 min to confirm death.
- X-ray-based verification
Table 1. OA cartilage histopathology grade assessment. Grade is depth progression into cartilage. Total score = Grade x Staging. 0 for normal joints, 24 for severe arthritis. Please click here to download this Table.
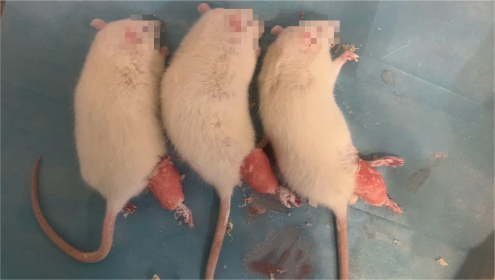
Figure 1. Rats immobilized in plaster. After the rats were anesthetized, their right lower limbs were wrapped with plaster bandages, fixed in the hyperextended position, and covered with a layer of denture base materials outside. Please click here to view a larger version of this figure.
3. Tuina manipulation
- Application area
- Select a total of 5 acupoints on the right hind limb of rats, including ST34, ST35, SP10, EX-LE4 and BL40 (Figure 2). Locate the acupoints according to the principles of acupoint positioning in 24.
- Position for application
- Cut a black cloth into a 9 cm x 15 cm bag with one side opening and tighten the opening with a rope. Before the Tuina manipulation, gently pull the rat's tail to make it burrow into the bag and expose its hind limbs outside the bag.
- Use one hand to keep the rat in the prone position, holding its tail and hind leg while the other hand applies press and kneading manipulation on a specific acupoint (Figure 3).
- Tuina manipulation
- Operate press and kneading manipulation on the 5 acupoints on the right hind limb for 2 min each. Place the performers thumb on the selected acupuncture point to perform rhythmic press and kneading, driving the skin and subcutaneous tissue together in a circular motion.
- Use finger pressure recordings (units in Newton) to ensure consistent intensity and frequency of the manipulation. Keep the intensity between 3-5 N and the frequency at 2 Hz (Figure 4). Apply the manipulation once a day for 21 days.
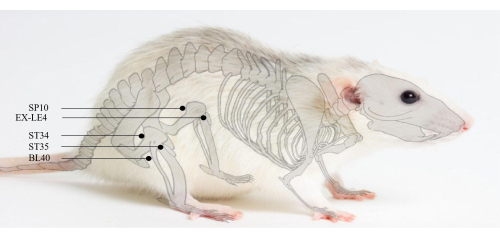
Figure 2. Acupoints position. SP10 is located 5 mm above the inner knee joint in rats. ST34 is located 5 mm above the outer knee joint in rats. EX-LE4 is located in the medial side of the knee ligament in rats. ST35 is located in the lateral side of the knee ligament in rats. BL40 is located at the midpoint of the transverse popliteal stripe. Please click here to view a larger version of this figure.
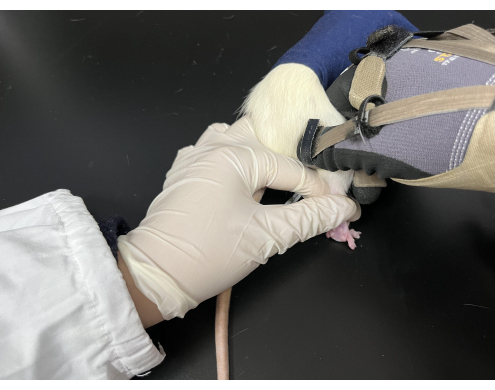
Figure 3. Tuina manipulation applied on rats. The rats were kept in a black bag with their hind limbs exposed. The performer held the rat's tail with the left hand while the right hand performed the manipulation. Please click here to view a larger version of this figure.

Figure 4. Finger pressure recordings. A device that records the force and frequency of finger pressure is used for real-time feedback on the intensity and frequency in the process of Tuina manipulation. (A) Pressure sensor and transmission equipment. (B) Finger pressure recordings. (C) The force recorded during Tuina manipulation. Please click here to view a larger version of this figure.
4. Pain Behavior Tests
- Test pain behavior before and after modeling, and at 1 day after (D1), 7 days after (D7), 14 days after (D14) and 21 days after (D21) the Tuina manipulation, including mechanical withdrawal threshold and paw withdrawal latency tests.
- Mechanical withdrawal threshold (MWT)
- Place the rats in a 20 cm x 10 cm x 20 cm transparent tempered glass cubicle located on a 40 cm high stage which is made up of a wire lattice with a size of 10 mm x 10 mm aperture. Keep the room temperature at 23 °C ± 2 °C.
- Settle the rats in the behavioral laboratory for at least 2 h per day during the behavioral test phase to avoid interference with the test results due to the animals' lack of adaptation to the environment at the beginning of the formal test. Place the rats in the behavioral laboratory for 30 min before the start of the formal test to facilitate their adaptation to the environment and to reduce distracting factors.
- Use electronic Von Frey fibers to measure the MWT. Stimulate the rat with the fiber at the center of its foot and withdraw the fiber when the rat shows obvious movements such as raising the leg and avoiding. The machine can automatically record the maximum pressure value (N) at this moment.
- Start the next stimulation in the same rat at least 15 s after the current stimulation. Do not exceed 5 s during each stimulation to prevent sensitization to tactile stimuli in the paws of rats. Repeat the test 5x until the difference between the three consecutive measurements is insignificant (within 10 N).
- Delete the values with large differences (the maximum and the minimum values) and take the average of the remaining three values as the mechanical withdrawal threshold.
- Paw withdrawal latency (PWL)
- Place the rats in a small compartment of transparent tempered glass with a size of 20 cm x 10 cm x 20 cm. Cover the top of the compartment with a transparent glass cover with ventilation holes. Keep the temperature of the transparent glass plate at 28-30 °C, on which the compartment is placed.
- Settle the rats in this environment for at least 30 min to acclimatize before the start of each formal test. If rats urinate or defecate in the compartment, clean it with absorbent paper in time to avoid affecting the subsequent light radiation heat transfer.
- Focus the spotlight on the center of the rat's foot and press the Start button. Press the Stop button when the rat shows obvious behaviors such as foot retraction or paw licking and record the time at this point. The spotlight irradiation time should not exceed 20 s to avoid damage to the rat's skin.
- Perform the next irradiation on the same rat after at least 10 min to prevent sensitization. Measure 5x on each rat.
- Remove the values with large differences (the maximum and the minimum value). Take the average of the remaining values as the PWL.
5. Sample preparation
- Perform euthanasia on the mouse as described in 2.3.2.1.
- Synovial membrane preparation
- Fix the rat in the supine position with a syringe needle on a foam board with the right hind limb flexed in the abduction and external rotation. Pinch up the skin around the knee joint with surgical scissors and expose the muscles around the knee joint of rats by cutting the skin and then cutting the subcutaneous fascia.
- Take the patellar ligament as a landmark to strip the muscle groups above the patellar ligament. Incise the ending of the patellar ligament (at the tibial tuberosity) carefully and find the synovial membrane when the ligament is pinched upwards from below.
- Carefully cut off the synovial tissue with ophthalmic scissors and wash off the blood and synovial fluid with pre-cooled saline. Fix the synovial membrane in 4% paraformaldehyde for at least 48 h after absorbing water from the tissue surface with clean gauze.
- Prepare the sample as in steps 2.3.2.5- 2.3.2.6. Perform scoring as in step 2.3.2.7.
- Performing hematoxylin and eosin staining
- Dewax to water with xylene for 20 min, replace with another round of fresh xylene for 20 min, followed by treating with anhydrous ethanol for 5 min, replace with another round of fresh anhydrous ethanol for 5 min, then add 90% ethanol by volume for 5 min, 80% ethanol by volume for 5 min, 70% ethanol by volume for 5 min, and finally distilled water for 5 min.
- Immerse the slide in hematoxylin staining solution for 3-8 min. Remove the slide and rinse off the stain with distilled water. Move it into the differentiation fluid (1% hydrochloric acid alcohol) for almost 30 s, so that the slide fades to pale blue. Rinse it with distilled water, and place in eosin staining solution for 1-3 min.
- Dehydrate the slide with 95% ethanol volume fraction for 5 min, replace with another round of fresh 95% ethanol for 5 min, followed by anhydrous ethanol for 5 min, replace with another round of fresh anhydrous ethanol for 5 min.
- Make the slide transparent by adding xylene for 5 min, replace it with another round of fresh xylene for 5 min, and then seal it with neutral resin.
- Terminal-deoxynucleotidyl transferase-mediated nick end labeling (TUNEL) on cartilage
- Dehydrate the cartilage sample in anhydrous ethanol, 90% ethanol, 85% ethanol and 75% ethanol until the dewaxing hydration is complete. Soak in PBS for 5 min. Add 3% H2O2 dropwise for 10 min.
- Add proteinase K working solution dropwise and digest at 37 °C for 10 min. Add 20 µL of labeling buffer per slice to keep it moist and shake off excess liquid after preparing the working solution. Add 20 µL of working solution to each slide and incubate for 2 h at 37 °C in a wet box.
- Add 50 µL of the closure solution drop by drop and close for 30 min. Then add 50 µL of diluted biotinylated anti-digoxin antibody (1:100 dilution) dropwise and incubate at 37 °C for 2 h in a wet box. Add 10 µL of SABC antibody diluent (1:100 dilution) dropwise and incubate at 37 °C for 2 h in a wet box.
- Add DAB color development solution (50 µL each of reagents A, B and C in 1000 µL of distilled water) dropwise for 10-15 min. The color development completes when it is brownish-yellow granular.
- Re-stain with hematoxylin for 3 s. After gradient dehydration and transparent treatment, dry at room temperature and seal the slide carefully with neutral gum. Pay attention to avoid leaving bubbles and overflowing glue.
- Immunohistochemical analysis of IL-1β and TNF-α
- Dewax the slides routinely in xylene and hydrate them in gradient alcohol. Inactivate the endogenous peroxidase in the sections with 3% H2O2. Place the slide holder in 95 °C citrate buffer (pH 6.0) and incubate in a water bath above 95 °C for over 20 min. Take out the incubation box and leave it at room temperature for at least 20 min.
- Incubate 5% normal goat serum with PBS for 10 min at 37 °C and shake off excess liquid. Add 150 µL of antibody I drop by drop and let stand at 37 °C for 1 h, then store overnight at 4 °C. Next day, re-warm at 37 °C for 45 min.
- Wash 3x with PBS for 5 min each. Cover the tissue on the slide with 3% BSA and seal it at 37 °C for 30 min. Add 150 µL of antibody II drop by drop and let stand at room temperature for 1 h. Wash 3x with PBS for 5 min each and add 150 pL of DAB color development liquid dropwise. Observe the degree of staining under the microscope until the sample becomes brownish-yellow even to the naked eye.
- Immediately, rinse with PBS for 10 min. Stain again with hematoxylin, dehydrate in gradient alcohol, make slides transparent in xylene, and seal with neutral gum.
- Statistical analysis
- Immunohistochemically stained sections with positive expression of the associated protein are yellow or brownish-yellow. Score the intensity of positive expression by Image J software for each group of immunohistochemical sections and use the evaluation criteria of average optical density (AOD), calculated by IOD divided by area.
- Use analysis software for statistical analysis. Use t-test if the data conformed to normal distribution and chi-square and use non-parametric test if they did not conform to normal distribution. Analyze repeated measurement data by generalized estimating equations.
Results
Pain behavior tests
The MWT results showed that the MWT of the right hind limb after modeling was significantly lower than before (p<0.05). Compared with the control group, the MWT of rats was significantly elevated after Tuina (p<0.05; Figure 5 and Table 2).
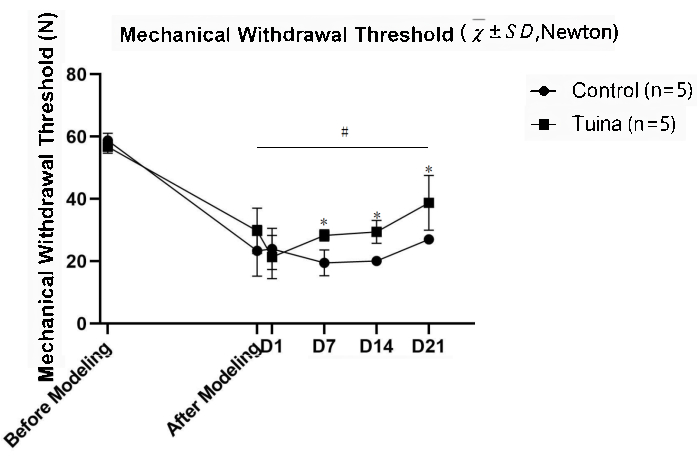
Figure...
Discussion
This study provides a protocol for Tuina manipulation on KOA rats. Through pain behavior tests and histomorphological findings, it suggested that such a series of Tuina manipulation applied to KOA rats could reduce synovial inflammation and cartilage apoptosis, which could be a reference of Tuina manipulation on animal models of KOA.
There are several critical procedures during the protocol. First, it's important to choose an appropriate method for inducing the KOA model. There are various...
Disclosures
The authors declare that they have no competing interests.
Acknowledgements
This work was supported by Shanghai Critical Clinical Specialties Construction Project (Grant Number: Shslczdzk04001); the Sailing program of Shanghai Science and Technology Commission (Grant Number: 22YF1444300); Projects within the budget of Shanghai University of Traditional Chinese Medicine (Grant Number: 2021LK091).
Materials
| Name | Company | Catalog Number | Comments |
| absolute ethanol | Supelco | PHR1070 | For making specimen |
| ALMEMO admeasuring apparatus | ahlborn | 2450-1 | For Mechanical Withdrawal Threshold test |
| Anti-Digoxin antibody | Sigma-Aldrich | SAB4200669 | For HE stain IHC or TUNEL |
| Anti-IL-1 beta | abcam | ab283818 | For HE stain IHC or TUNEL |
| DAB Substrate kit | Solarbio | DA1010 | For HE stain IHC or TUNEL |
| Denture base materials | Shanghai New Century | 20000356 | For model making |
| eosin | bioswamp | PAB180016 | For HE stain IHC or TUNEL |
| Finger pressure recordings | Suzhou Changxian Optoelectronic Technology | CX1003w | For Tuina manipulation |
| formic acid solution | Sigma-Aldrich | 695076 | For decalcification |
| H2O2 | Sigma-Aldrich | 386790-M | For HE stain IHC or TUNEL |
| hematoxylin | bioswamp | PAB180015 | For HE stain IHC or TUNEL |
| Isoflurane | Shanghai Yuyan Scientific Instrument Company | S10010533 | For gas anesthesia |
| neutral resins | bioswamp | PAB180017 | For HE stain IHC or TUNEL |
| Paraformaldehyde Fix Solution | Sigma-Aldrich | 100496 | For histology |
| PBS | Sigma-Aldrich | P3813 | For HE stain IHC or TUNEL |
| Plantar Test Apparatus | IITC Life Science | / | For Paw Withdrawal Latency test |
| plaster of Paris bandage | WANDE | 20150023 | For model making |
| Proteinase K | Sigma-Aldrich | 124568 | For HE stain IHC or TUNEL |
| TNF Alpha Monoclonal antibody | Proteintech | 60291-1-Ig | For HE stain IHC or TUNEL |
| TUNEL | Servicebio | GDP1042 | For HE stain IHC or TUNEL |
| Wax | Sigma-Aldrich | 327204 | For making specimen |
| xylene | Shanghai Sinopharm Group | 100092 | For making specimen |
References
- Joint Surgery Group of Chinese Orthopaedic Association, Chinese Association of Orthopaedic Surgeons, National Clinical Research Center for Geriatric Diseases, Chinese Journal of Orthopaedics. Chinese Osteoarthritis Treatment Guidelines (2021 Edition). Chinese Journal of Orthopaedics. 41 (18), 24 (2021).
- David, S., et al. Epidemiology of knee osteoarthritis in general practice: a registry-based study. BMJ Open. 10 (1), 031734 (2020).
- Callahan, L. F., Cleveland, R. J., Allen, K. D., Golightly, Y. Racial/Ethnic, Socioeconomic, and Geographic Disparities in the Epidemiology of Knee and Hip Osteoarthritis. Rheumatic Disease Clinics of North America. 47 (1), 1-20 (2021).
- Wang, K., Dong, X., Lin, J. H. Investigation of Medical Costs of Disease In Patients With Osteoarthritis of the Knee Joint. National Medical Journal of China. 97 (1), 4 (2017).
- Perlman, A., et al. Efficacy and Safety of Massage for Osteoarthritis of the Knee: a Randomized Clinical Trial. Journal of General Internal Medicine. 34 (3), 379-386 (2019).
- Xing, H., et al. Therapeutic massage for knee osteoarthritis: a systematic review and meta-analysis of randomized controlled trials. Journal of Acupuncture and Tuina Science. 19 (5), 354-363 (2021).
- Seo, B. R., et al. Skeletal muscle regeneration with robotic actuation-mediated clearance of neutrophils. Science translational medicine. 13 (614), (2021).
- Wu, J. H., Zhang, C., Dong, S. J., Yin, H. Effects of Massage with #34;Relaxing Tendons" Technique on the Interleukin-1B and 5-hydroxytryptamine Levels in the Joint Fluid of a Rabbit Knee Osteoarthritis Model. Chinese General Practice. 21 (6), 688-693 (2018).
- Luo, R. The effects of IL-1B,IL-6,IL-13,IL-26,TNF-a by patellar manipulations on rabbit knee osteoarthritis model. Guangxi University of Traditional Chinese Medicine. , (2017).
- Qiu, F., Li, C., Wu, X., Liu, Y., Zhang, X. Effect of Massage on the Function of Foot-Yangming Meridian-Muscle in Patients with Knee Osteoarthritis. Acta Chinese Medicine. 36 (3), 649-655 (2021).
- Yang, B., Li, S. Research Progress of Massage in Improving Biomechanical Indexes of Knee Osteoarthritis. Acta Chinese Medicine. 37 (12), 2571-2576 (2022).
- Li, C., Qiu, F., Ding, J., Hu, G., Zhang, X. Curative Observation of Retaining of Heated Needle at Trigger Points Combined with Manipulation for Muscles along Meridians in Treating Knee Osteoarthritis. Journal of Guangzhou University of Traditional Chinese Medicine. 37 (11), 2157-2162 (2020).
- Ding, X., Zhang, X., Hou, Y., Zhao, Z., Ye, X. Effect of massage manipulation on joint stiffness of knee osteoarthritis patients based on "spine-pelvis-knee" holistic diagnosis and treatment pattern. Shanghai Journal of Traditional Chinese Medicine. 55 (08), 54-57 (2021).
- Fu, Y., Gong, L., Li, Y. The clinical studies of Rolling combined with Pulling Manipulation on Early and Middle-term. Jilin Journal of Chinese Medicine. 40 (07), 958-962 (2020).
- Jiang, J., Hu, X., Tang, R., Qiu, F., Huang, L. Change of lower limb force line in treating knee osteoarthritis with manipulation. Journal of Changchun University of Chinese Medicine. 34 (01), 129-132 (2018).
- Liang, Y., et al. Some issues on animal experiments standardization of acupuncture and moxibustion. Lishizhen Medicine and Materia Medica Research. 25 (09), 2299-2300 (2014).
- Guo, Y., Liu, Y., Liu, Q., Chen, Z., Zhao, X. Basic System of Chinese Acupuncture Standard. Chinese Acupuncture & Moxibustion. 31 (6), 549-550 (2011).
- YAN, X., Yan, J. Study on the Standardization of Classification of Tuina Manipulation. Acta Chinese Medicine. 32 (5), 875-878 (2017).
- Aikebaier, G., Lu, X., Liu, J., Liu, l., Wang, S. Analysis on Manipulation and Acupoint Selection Laws of Massage for Treatment of Knee Osteoarthritis Based on Data Mining Technology. Chinese Journal of Information on Traditional Chinese Medicine. 29 (5), 23-29 (2022).
- Gong, L., Wuquan, S., Zhang, H., Chen, Z. Research of Yan Juntao's Academic Experiences of Differential Treatment and Manipulation for Treating of Knee Osteoarthritis. Chinese Journal of Traditional Medical Traumatology & Orthopedics. 24 (7), 16-19 (2016).
- Qian, J., Xing, X., Liang, J. Two Methods to Establish Rat Model of Osteoarthritis of the Knee. Research and Exploration in Laboratory. 33 (11), 23-27 (2014).
- Liu, J., et al. Experimental study of a modified Videman method for replicating knee osteoarthritis on rabbit. Rehabilitation Medicine. 30 (03), 212-219 (2020).
- Moskowitz, R. W. Osteoarthritis cartilage histopathology: grading and staging. Osteoarthritis and Cartilage. 14 (1), 1-2 (2005).
- Shen, M., Li, Z., Shen, J. Preliminary Exploration of Experiment Teaching on Experiment Acupuncture Science. Chinese Medicine Modern Distance Education of China. 7 (02), 130-131 (2009).
- Jeong, J., et al. Anti-osteoarthritic effects of ChondroT in a rat model of collagenase-induced osteoarthritis. BMC complementary and alternative medicine. 18 (1), 131 (2018).
- Hulth, A., Lindberg, L., Telhag, H. Experimental osteoarthritis in rabbits. Preliminary report. Acta orthopaedica Scandinavica. 41 (5), 522-530 (1970).
- Tawonsawatruk, T., Sriwatananukulkit, O., Himakhun, W., Hemstapat, W. Comparison of pain behaviour and osteoarthritis progression between anterior cruciate ligament transection and osteochondral injury in rat models. Bone & Joint Research. 7 (3), 244-251 (2018).
- Zhou, Q., et al. Cartilage matrix changes in contralateral mobile knees in a rabbit model of osteoarthritis induced by immobilization. BMC musculoskeletal disorders. 16, 224 (2015).
- Zeng, J., et al. Establishment and identification of experimental rabbit model of knee osteoarthritis. Chinese Journal of Clinical Research. 29 (5), 679-682 (2016).
- Shang, P., et al. Comparison with two kind osteoarthritis animal models reduced by plaster immobilization in extend excessive position and bend excessive position respectively. Orthopaedic Biomechanics Materials and Clinical Study. (1), 11-14 (2006).
- He, Y., et al. Evaluation of the effect of improved cast immobilization method on rabbit knee osteoarthritis model. Chinese Imaging Journal of Integrated Traditional and Western Medicine. 18 (2), 198-219 (2020).
- Pengfei, S., et al. Possible mechanism underlying analgesic effect of Tuina in rats may involve piezo mechanosensitive channels within dorsal root ganglia axon. Journal of Traditional Chinese Medicine. 38 (6), 834-841 (2018).
- Wang, Y. Mechanisms of Massage Mediating Chondrocyte Apoptosis in Knee Osteoarthritis Through Piezo 1/JAK2 Signaling Pathway. Shandong University of Traditional Chinese Medicine. , (2019).
- Liu, J. Effect of Massage on TLR4/MyD88 Signal Transduction Pathway in Rat Knee Osteoarthritis Model. Guangzhou University of Traditional Chinese Medicine. , (2019).
- Yu, S., Zhou, J., Pang, X. Advances in induced animal models of knee osteoarthritis. Journal of Guangxi University of Chinese Medicine. 25 (5), 50-55 (2022).
- Tan, Q., et al. Acupuncture combined with moxibustion regulates the expression of circadian clock protein in the synovium of rats with osteoarthritis. Chinese Journal of Tissue Engineering Research. 26 (11), 1714-1719 (2022).
- Zhang, Z., et al. A Review of Massage Therapy for Knee Osteoarthritis. Henan Traditional Chinese Medicine. 39 (1), 146-149 (2019).
- Gong, L., Sun, W., Zhang, H., Chen, Z. Research of Yan Juntao's Academic Experiences of Differential Treatment and Manipulation for Treating of Knee Osteoarthritis. Chinese Journal of Traditional Medical Traumatology & Orthopedics. 24 (7), 16-19 (2016).
- Chen, C., Zhang, H. Research on the rules of acupoint selection for the treatment of knee osteoarthritis with massage based on data mining. Hainan Medical Journal. 29 (18), 2617-2619 (2018).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved