A subscription to JoVE is required to view this content. Sign in or start your free trial.
Application of Acupotomy in a Knee Osteoarthritis Model in Rabbit
In This Article
Summary
In this protocol, a knee osteoarthritis model was prepared using the modified Videman method, and the operation procedures and precautions of acupotomy are detailed. The effectiveness of acupotomy has been demonstrated by testing the mechanical properties of quadriceps femoris and tendon and the mechanical and morphological properties of cartilage.
Abstract
Knee osteoarthritis (KOA) is one of the most frequently encountered diseases in the orthopedic department, which seriously reduces the quality of life of people with KOA. Among several pathogenic factors, the biomechanical imbalance of the knee joint is one of the main causes of KOA. Acupotomology believes that restoring the mechanical balance of the knee joint is the key to treating KOA. Clinical studies have shown that acupotomy can effectively reduce pain and improve knee mobility by reducing adhesion, contracture of soft tissues, and stress concentration points in muscles and tendons around the knee joint.
In this protocol, we used the modified Videman method to establish a KOA model by immobilizing the left hindlimb in a straight position. We have outlined the method of operation and the precautions related to acupotomy in detail and evaluated the efficacy of acupotomy in conjunction with the theory of "Modulating Muscles and Tendons to Treat Bone Disorders" through the detection of the mechanical properties of quadriceps femoris and tendon, as well as cartilage mechanics and morphology. The results show that acupotomy has a protective effect on cartilage by adjusting the mechanical properties of the soft tissues around the knee joint, improving the cartilage stress environment, and delaying cartilage degeneration.
Introduction
Knee osteoarthritis (KOA) is the most frequent form of osteoarthritis, often recognized as a whole-joint disease characterized by articular cartilage degeneration, which manifests clinically as pain, swelling, and limited movement of the affected joints1. According to recent epidemiological statistics, KOA is reported to have affected 654.1 million individuals globally who were 40 years of age or older by 2020. The prevalence and incidence of KOA rise with age, are the highest in middle-aged and older adults, and affect more women than men2. The prevalence of KOA is likely to increase due to the aging population and obesity epidemic worldwide, posing a growing threat to global public health. Age, sex, obesity, trauma, and other complicated risk factors associated with KOA all directly impact knee instability, making a biomechanical imbalance in knee joints one of the primary causes of KOA3.
Under normal physiological conditions, the knee joint is in a state of mechanical balance, ensuring that the mechanical loads in the joint are evenly distributed on the cartilage. Any mechanical imbalance in the knee joint can lead to abnormal stress in cartilage, resulting in cartilage degeneration and the onset of KOA4. The muscle-tendon system is the main dynamic system that maintains the mechanical balance of the knee joint. The coordinated movement of the extensor and flexor muscle-tendon system can evenly distribute the load generated by the movement on the cartilage surface, avoiding the metabolic imbalance of local cartilage stresses beyond its physiological load that results in cartilage loss5. Decreased muscle strength is the main cause of intramuscular movement disorder and cartilage damage, which may occur before symptomatic KOA.
KOA can also induce arthrogenous muscle inhibition (AMI), manifesting as muscle weakness and decreased muscle strength around the knee6. Among these muscles, the quadriceps femoris group functions as the only knee extensor, an important structure in maintaining knee joint stability. Studies have shown that a decrease in quadriceps cross-sectional area and muscle strength is significantly and positively correlated with KOA progression7. The decline in quadriceps strength affects the gait pattern, knee stability, movement patterns, and many other functions. Moreover, the decline in muscle strength impairs tendon function, manifested as a decrease in tendon stiffness, elastic modulus, and other biomechanical properties8. In long-term strain repair, changes such as adhesion and contracture may occur in the muscles and tendons of the knee joint, damaging their mechanical properties, causing joint instability, and ultimately forming a vicious cycle of pathological changes of KOA. It is, therefore, crucial for KOA treatment to improve the mechanical properties of the muscle-tendon system and restore the joint mechanical balance.
Among the causes of KOA, biomechanical imbalance is the main inducing factor for knee pain, dysfunction, inflammatory lesions, and cartilage degeneration9. Therefore, the key to treating KOA is to restore the biomechanical balance of the knee joint. Acupotomology believes that the etiology and pathogenesis of KOA are "mechanical imbalance." When the mechanical characteristics of the soft tissues around the knee change abnormally, the knee joint loses its mechanical balance, and the abnormal mechanical stress environment of the joint accelerates degeneration, causing inflammatory stimulation to further aggravate the soft tissue adhesions, contractures, and further decline in joint stability. This vicious cycle eventually develops into KOA. By loosening soft tissue adhesions and contractures, as well as reducing stress concentration in the muscles and tendons, acupotomy in conjunction with the theory of "Modulating Muscles and Tendons to Treat Bone Disorders" improves the soft tissue mechanics and "modulates muscles and tendons," which balances the mechanical stress of the joint, effectively alleviating cartilage degeneration and "treating bone disorders"10. In terms of animal model selection, based on the purpose of this study, we prepared the KOA model by the modified Videman method of left hindlimb extension immobilization.
This paper details the establishment of the KOA model using the modified Videman method of left hind limb extension immobilization and the method of operation and precautions of acupotomy. We demonstrate the effectiveness of acupotomy by testing the mechanical properties of quadriceps femoris and tendon and detecting changes in articular cartilage stress and morphology.
Protocol
All animal experiments were reviewed and approved by the Animal Ethics Committee of Beijing University of Chinese Medicine (No. BUCM-4-2022010101-1097). In this protocol, 24 6-week-old male New Zealand rabbits were housed under a specific condition, namely, 20-25 °C, 50-60% humidity, and a 12 h light/12 h dark circadian cycle, with free access to a regular chow diet. The rabbits were anesthetized and sacrificed by combining deep anesthesia and air embolization. Pain is one of the typical pathological features of KOA and is also one of the key indicators used to evaluate animal models of KOA and intervention methods, so analgesics are not used during model preparation.
1. The KOA rabbit model
- Anesthetize rabbits with 3% pentobarbital sodium (30 mg/kg) intravenously at the ear margin. To confirm the appropriate level of anesthesia, look for a significantly weakened or absent corneal reflex and the absence of pain upon clamping the skin with hemostatic forceps. During anesthesia, add 2-3 drops of lubricant to the rabbits' eyes every 15 min to prevent the rabbits' eyes from drying out.
- After anesthesia, fix each rabbit in the supine position, pulling the left hindlimb into a fully extended position.
- Fix the left hind limb of each rabbit in the extended position.
- As the first layer, use medical tape to cover the rabbit's skin from the groin to the ankle joint.
- As the second layer, wrap 36 mm-wide, double-sided foam tape over the medical tape and then wrap a polymer bandage from the groin to the ankle joint. Ensure that the knee joint is 180° straight and the ankle joint is dorsiflexed by 60°.
- As the third layer, immobilize the joints with small splints at the front and back of the knee and ankle joints, and wrap steel mesh around the outermost layer to protect against bites. Expose the rabbits' toes to observe if the blood circulation is normal.
- Immobilize the animals for 6 weeks to establish the KOA model (Figure 1).
NOTE: 1) During model preparation, inspect the molds every other day. If any molds are loose or detached, anesthetize the rabbits, and re-immobilize the left hind limbs in an extended position. 2) Lay protective mats on the bottom of the cages to prevent the rabbits' limbs from getting stuck and causing injury.
2. Acupotomy intervention
NOTE: Before the start of acupotomy intervention, anesthetize the rabbits with 3% pentobarbital sodium (30 mg/kg) by ear margin intravenous injection.
- Determine the points of treatment.
- Shave off the fur of the knee joint of the rabbit's left hind limb.
- Palpate the rabbit knee joint medial femoral muscle tendon insertion, rectus femoral tendon insertion, biceps femoris tendon insertion, and goosefoot bursa. Mark pathological indurations of local muscles with a sterile skin marker. Disinfect the knee joint three times with alternating rounds of medical iodophor and 75% medical alcohol.
- Operation of acupotomy
- Keep the acupotomy blade parallel to the direction of travel parallel to the tendon and the longitudinal axis of the limb.
- Use the thumb of the left hand to press down on the skin entering the marker point and move laterally so that blood vessels and nerves are separated on the ventral side of the thumb.
- With the acupotomy handle in the right hand, press down quickly with a small force so that the acupotomy blade instantly passes through the skin. Slowly advance the acupotomy blade to the local muscle indurations and make longitudinal cuts and swings laterally.
- After the acupotomy operation is complete, disinfect the knee joint again and apply a band-aid.
- Perform this operation once a week for 4 weeks (Figure 2).
NOTE: 1) If there is no induration or cord-like tissue touched at the tendon insertion of the vastus medialis, vastus lateralis, rectus femoris, biceps femoris, or the anserine bursa, the acupotomy needle should be used to release their tendon insertions directly. 2) During acupotomy intervention, do not immobilize the left hind limbs of rabbits in the acupotomy group and the model group in extension position.
3. Elastic modulus of quadriceps femoris
NOTE: 1) This experiment used the real-time shear wave elastography (SWE) ultrasound diagnostic instrument to measure the elastic modulus of the quadriceps femoris in vivo in each group of rabbits. 2) The tester should be an experienced sonographer in ultrasound detection. When measuring, the ultrasound probe should be gently placed on the skin surface of the quadriceps to avoid local muscle tension. Measurements need to be taken when the animal is in a quiet state, without struggling or activity. If the animal is active, wait until it is calm before conducting the test.
- Shave the fur to expose the skin in the quadriceps region of the left hindlimb.
- Use conventional two-dimensional ultrasound to locate the quadriceps musculo-abdomen and determine the region of interest (ROI), set to a depth of 1-2 cm.
- Start the SWE mode for inspection.
- Set the area of interest uniformly to a circular area with a diameter of 2 mm and the area of interest to ~0.5-1 cm deep from the surface of the skin.
- Use the ultrasonic diagnostic instrument to generate an acoustic radiation force impulse to stimulate the muscle tissue and obtain tissue elastography.
- Wait for 2-3 s for the image to stabilize and then freeze the image. Activate the Q-BOX function of the instrument to measure the Young's modulus of the quadriceps muscle.
- Wait for the system to automatically calculate the maximum, minimum, and average values (unit: KPa) of Young's modulus of the ROI. Select three ROIs at the same depth for three measurements and take the average value for statistical analysis.
NOTE: The tester should be an experienced sonographer in ultrasound detection. When measuring, the ultrasound probe should be gently placed on the skin surface of the quadriceps to avoid local muscle tension. Measurements need to be taken when the animal is in a quiet state, without struggling or activity. If the animal is active, wait until it is calm before conducting the test.
4. Measuring the contraction force of quadriceps femoris
NOTE: After the measurement of the contraction force of the quadriceps femoris, the rabbits were euthanized by air embolism under anesthesia.
- Anesthetize the rabbits with 3% pentobarbital sodium (30 mg/kg) intravenously at the ear margin. To confirm that the appropriate level of anesthesia has been reached, look for a significantly weakened or absent corneal reflex and the absence of pain upon clamping the skin with hemostatic forceps. During anesthesia, add 2-3 drops of lubricant to the rabbits' eyes every 15 min to prevent the rabbits' eyes from drying out.
- Expose the quadriceps muscles and attach the tension transducer.
- Cut the skin below the patella, along the longitudinal axis of the limb upwards to the base of the thigh, and continue to cut the skin upwards by 3-4 cm. Carefully peel off the skin and fascia, and expose the muscle. Cut the patellar ligament and carefully separate the quadriceps from the iliac junction, keeping the quadriceps in connection with the iliacium.
- Ligate the surgical sutures at the tendon junction between the patella and the quadriceps muscle. Stretch the muscle to its full length in its natural state and then attach it to the tension transducer. Keep the ligation line on the muscle in a straight line with the ligation line on the force transducer.
- Secure the tension transducer to the bracket. Connect the signal acquisition line on the tension transducer to the biosignal acquisition system processor.
- Measure the contractile performance of the quadriceps muscle.
- Insert the electrodes parallel to the quadriceps abdomen and avoid any contact between the electrodes.
- Press the oscilloscope button. Adjust the position of the force transducer on the bracket to maintain the baseline at zero. Select the stimulation parameters of the stimulator with a wave width of 5 ms and a delay of 10 ms.
- Use a single stimulus first and gradually adjust the stimulus intensity from zero with an increment of 0.1 V each time. Observe the changes in the muscle contraction curve and contraction amplitude until the maximum single contraction amplitude (Pt) of the quadriceps is determined. Record it for subsequent statistics.
- Use a cluster stimulus, and use the stimulus amplitude that induces the maximum single contraction amplitude as the baseline to continuously stimulate the muscle and gradually increase the stimulus frequency. Observe the changes in the muscle contraction curve until the maximum contraction amplitude (Pt) of the quadriceps is determined. Record it for subsequent statistics.
NOTE: 1) After each muscle contraction, the muscle should be given 30 s to relax with the muscle buffer solution being dripped continuously on the muscle. 2) During the operation, judge the anesthesia status by monitoring the rabbits' eyelid reflex, respiratory rhythm, muscle relaxation and skin pinching response.
5. The mechanical performance of the quadriceps tendon
- Preprocessing: On the day of testing, measure the length, width, and thickness of the quadriceps tendon with a vernier caliper, and install a special anti-slip clamp in the fatigue testing machine. Repeat loading and unloading 15x for preprocessing.
- Stress relaxation test: Use the sensor ranging from 0 N to 100 N, stretch it at a speed of 5 mm/min until it reaches the required length, and then start collecting data. Set the computer data acquisition time from t (0), collecting data every 0.1 s, lasting for 1,800 s. After reaching the set time, record data and curves.
- Tensile test: Use the sensor ranging from 0 N to 100 N and stretch it at a speed of 5 mm/min to the maximum load until the specimen is pulled apart. After the test, calculate the maximum displacement, ultimate load, and stiffness of the specimen.
6. Joint contact surface pressure and pressure per unit area of the cartilage
- Fix the femur and tibia specimens on both sides in a straight position on the fixture and perform a preload test. Measure the approximate range of the knee joint, cut the pressure-sensitive paper into the same shape, and seal it with plastic wrap.
- Place the sealed pressure-sensitive paper between the tibia and femur joints, and conduct a pressure test on the knee joint with a pressure of 5 mm/min and a maximum pressure of 50 N. Maintain the pressure for 2 min until it reaches 50 N when the pressure-sensitive paper is stably colored.
- After 2 min, remove the pressure-sensitive paper, fix the colored surface on an A4 size sheet of paper, and acquire images with the scale set aside.
- Upload the image to the computer. Use the referenced software for area measurement and multi-segment measurement for irregular figures. Measure the pressure on the inside and outside of the joints of the femur and tibia and record the results.
7. Safranin O/Fast Green staining of the knee joint cartilage
- After the end of the acupotomy intervention, take the cartilage-subchondral bone complex tissues and embed them in paraffin. Slice the prepared tissue wax blocks and prepare slides. Deparaffinize the prepared tissue slides with environmental dewaxing solution(I) and environmental dewaxing solution (II) for 15 min each; then, dip them successively in xylene and anhydrous ethanol (1:1), anhydrous ethanol (I), 95% ethanol, 85% ethanol, and 75% ethanol, 2-5 min each step; and finally, soak them in distilled water for 15 min.
- Perform staining.
- Stain the slides with Fast Green solution for 1 min. During this process, take the slides out of the solution and observe them under the microscope until the tissue is stained dark green.
- Color separation: Rinse the excess Fast Green solution with ultrapure water. Soak the slides quickly in 1% acetic acid solution for 5 - 10 seconds. Again, rinse the slide with ultrapure water.
- Stain the slides in Safranine O solution for 10-15 min until the cartilage is stained red.
- Dehydrate and clarify the tissue, seal the glass slides, and observe them under the microscope.
- Soak the slides in 75% ethanol, 85% ethanol, 95% ethanol, and 100% ethanol for 3 - 5 seconds successively.
- Dip the slides in environmental dewaxing solution(I) and environmental dewaxing solution (II) for 10 min successively. Take out the slides and drop the neutral resinous medium on the front of the slides, avoiding the tissue. Place the edge of the coverglass on the slide and then, slowly put it down to cover the neutral balsam. Remove the air and avoid air bubbles. Wipe off the extra xylene and neutral balsam, and let it stand overnight at room temperature.
- Observe the slides under the microscope and acquire images. For each group, select six samples of rabbit knee cartilage and randomly select four different viewing fields for each sample for evaluation. Score the cartilage histology of each group according to the Mankin method (Table 1).
8. Statistical analysis
- Express data as mean ± standard deviation (
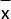 ± s).
± s). - Perform one-way analysis of variance (ANOVA) and LSD's test for determining the statistical significance of multiple group comparisons.
- Consider differences statistically significant when P < 0.05.
Results
Experimental results of mechanical properties of quadriceps femoris and tendon
To evaluate the effect of acupotomology on the mechanical properties of quadriceps femoris in rabbits with KOA, we used real-time shear wave elastic ultrasound imaging and a muscle tension transducer, respectively. Compared with the control group, Young's modulus of the quadriceps femoris in the KOA group was decreased (P < 0.05). Compared with the KOA group, Young's modulus of the acupotomy group wa...
Discussion
An appropriate animal model is one of the key factors to achieve experimental objectives and clarify a specific scientific question. This study was based on the theories of "Zongjin controlling bones and lubricating joints" and "mechanical imbalance" in acupotomology, aiming to explain the scientific connotation behind the treatment of KOA by "modulating muscles and tendons to treat bone disorders" in acupotomy therapy. In other words, acupotomy improves the abnormal mechanical environment of the ...
Disclosures
The authors have no conflicts of interest to disclose.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No.82074523,82104996).
Materials
| Name | Company | Catalog Number | Comments |
| Acupotomy | Beijing Zhuoyue Huayou Medical Devices Co., Ltd. | 0.4 x 40 mm | |
| Connect Cast Orthopedic Casting Tape | Suzhou Connect Medical Technology Co.,Ltd. | KCP06 | 15.0 cm x 360 cm |
| Double-sided Foam Tape | Deli Group Co.,Ltd. | NO.30416 | 36 mm x 5 yard x 2.5 mm |
| Environmental Dewaxing Solution | Wuhan Servicebio Technology Co.,Ltd. | G1128 | |
| Ethanol absolute | Beijing Hengkangda Medicine Co., Ltd. | ||
| Fast Green solution | Wuhan Servicebio Technology Co.,Ltd. | G1031 | |
| Fast grenn FCF | Sigma,America | 2353-45-9 | |
| Fatigue testing machine | BOSE, America | Bose Electro Force 3300 | |
| Four-channel physiological recorder | Chengdu Instrumeny Frctory | RM-6420 | |
| FPD-305E | Fuji, Japan | ||
| FPD-306E | Fuji, Japan | ||
| Hematoxylin solution | Wuhan Servicebio Technology Co.,Ltd. | G1005 | |
| Medical iodophor disinfectant | Shan Dong Lircon Medical Technology Co., Ltd. | ||
| Medical Tape | Shandong Rongjian Sanitary Products Co., Ltd. | 200402 | 1.5 x 500 cm |
| Muscle tension transducer | Chengdu Instrumeny Frctory | JH-2204005, 50 g | |
| Prescale | Fuji, Japan | ||
| Real-time SWE ultrasound diagnostic instrument | SuperSonic Imagine SA,France | SuperSonic Imagine AixPlorer | |
| Rhamsan gum | Wuhan Servicebio Technology Co.,Ltd. | WG10004160 | |
| Safranine O | Sigma,America | 477-73-6 | |
| Safranine O solution | Wuhan Servicebio Technology Co.,Ltd. | G1015 | |
| Statistical Package for the Social Sciences (SPSS) | IBM, America |
References
- Roseti, L., Desando, G., Cavallo, C., Petretta, M., Grigolo, B. Articular cartilage regeneration in osteoarthritis. Cells. 8 (11), 1305 (2019).
- Cui, A. Y., et al. Global, regional prevalence, incidence and risk factors of knee osteoarthritis in population-based studies. EClinicalMedicine. 29-30, 100587 (2020).
- Miyauchi, A., et al. Alleviation of murine osteoarthritis by deletion of the focal adhesion mechanosensitive adapter, Hic-5. Sci Rep. 9 (1), 15770 (2019).
- Wright, T. Biomechanical factors in osteoarthritis: the effects of joint instability. HSS J. 8 (1), 15-17 (2012).
- Patsika, G., Kellis, E., Kofotolis, N., Salonikidis, K., Amiridis, I. G. Synergetic and antagonist muscle strength and activity in women with knee osteoarthritis. J Geriatr Phys Ther. 37 (1), 17-23 (2014).
- Blalock, D., Miller, A., Tilley, M., Wang, J. X. Joint instability and osteoarthritis. Clin Med Insights. Arthritis and Musculoskelet Disord. 8, 15-23 (2015).
- Henriksen, M., Christensen, R., Danneskiold-Samsøe, B., Bliddal, H. Changes in lower extremity muscle mass and muscle strength after weight loss in obese patients with knee osteoarthritis: a prospective cohort study. Arthritis Rheum. 64 (2), 438-442 (2012).
- Schwartz, A. G., Lipner, J. H., Pasteris, J. D., Genin, G. M., Thomopoulos, S. Muscle loading is necessary for the formation of a functional tendon enthesis. Bone. 55 (1), 44-51 (2013).
- Felson, D. T. Osteoarthritis as a disease of mechanics. Osteoarthr Cartil. 21 (1), 10-15 (2013).
- Ma, S. N., et al. Effect of acupotomy on FAK-PI3K signaling pathways in KOA rabbit articular cartilages. Evid Based Complement Alternat Med. 2017, 4535326 (2017).
- Yu, P., et al. Research progress of experimental animal models of knee osteoarthritis. China Medical Herald. 16 (27), 41-44 (2019).
- Li, L. H., et al. Research progress of rabbit knee osteoarthritis model. Journal of Jiangxi University of Chinese Medicine. 31 (4), 108-113 (2019).
- Zhang, W., Gao, Y., Guo, C. Q., Khattab, I. Z. A., Mokhtari, F. Effect of acupotomy versus electroacupuncture on ethology and morphology in a rabbit model of knee osteoarthritis. J Tradit Chin Med. 39 (2), 229-236 (2019).
- An, X. Y., et al. Chondroprotective effects of combination therapy of acupotomy and human adipose mesenchymal stem cells in knee osteoarthritis rabbits via the GSK3 beta-cyclin D1-CDK4/CDK6 signaling pathway. Aging Dis. 11 (5), 1116-1132 (2020).
- Guo, C. Q., Liu, N. G. Analysis on the distribution features of Ashi points in Jingjin disorders. Journal of Basic Chinese Medicine. 17 (8), 899-900 (2011).
- Guo, C. Q., et al. Effects of acupotomy therapy on tenderness point around knee joint in patients with knee osteoarthritis: a randomized controlled clinical trial. Journal of Chengdu University of Traditional Chinese Medicine. 33 (3), 3-5 (2010).
- Zhao, Y., Dong, F. H., Zhang, K. Analysis of soft tissues mechanical changes and treatment of meridian tendon pain. Beijing Journal of Traditional Chinese Medicine. (9), 705-707 (2008).
- Zhang, L. P., Cheng, F., Liu, D. Y., Zhu, L. G. Application progress of biomechanics in knee osteoarthritis. Chinese Archives of Traditional Chinese Medicine. 34 (7), 1644-1647 (2016).
- Thomas, A. C., Sowers, M., Karvonen-Gutierrez, C., Palmieri-Smith, R. M. Lack of quadriceps dysfunction in women with early knee osteoarthritis. J Orthop Res. 28 (5), 595-599 (2010).
- Li, L., et al. Effects of unloaded muscle atrophy on contractile characteristics of hind-limb skeletal muscles in mice. Space Med Med Eng. 25 (5), 322-325 (2012).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved