A subscription to JoVE is required to view this content. Sign in or start your free trial.
3D Bioprinting Phototunable Hydrogels to Study Fibroblast Activation
In This Article
Summary
This article describes how to 3D bioprint phototunable hydrogels to study extracellular matrix stiffening and fibroblast activation.
Abstract
Phototunable hydrogels can transform spatially and temporally in response to light exposure. Incorporating these types of biomaterials in cell-culture platforms and dynamically triggering changes, such as increasing microenvironmental stiffness, enables researchers to model changes in the extracellular matrix (ECM) that occur during fibrotic disease progression. Herein, a method is presented for 3D bioprinting a phototunable hydrogel biomaterial capable of two sequential polymerization reactions within a gelatin support bath. The technique of Freeform Reversible Embedding of Suspended Hydrogels (FRESH) bioprinting was adapted by adjusting the pH of the support bath to facilitate a Michael addition reaction. First, the bioink containing poly(ethylene glycol)-alpha methacrylate (PEGαMA) was reacted off-stoichiometry with a cell-degradable crosslinker to form soft hydrogels. These soft hydrogels were later exposed to photoinitator and light to induce the homopolymerization of unreacted groups and stiffen the hydrogel. This protocol covers hydrogel synthesis, 3D bioprinting, photostiffening, and endpoint characterizations to assess fibroblast activation within 3D structures. The method presented here enables researchers to 3D bioprint a variety of materials that undergo pH-catalyzed polymerization reactions and could be implemented to engineer various models of tissue homeostasis, disease, and repair.
Introduction
3D bioprinting is a transformative technology that enables researchers to precisely deposit cells and biomaterials within 3D volumes and recreate the complex hierarchical structure of biological tissues. Over the past decade, advances in 3D bioprinting have created beating human cardiac tissues1, functional models of kidney tissues2, models of gas exchange within the lung3, and tumor models for cancer research4. The invention of embedded 3D bioprinting techniques, such as Freeform Reversible Embedding of Suspended Hydrogel (FRESH) bioprinting, has made it possible to reproduce complex soft tissue structures such as pulmonary blood vessels5 and even human heart6 in 3D. FRESH 3D bioprinting facilitates layer-by-layer printing of soft and low-viscosity bioinks through extrusion into a shear-thinning support bath. The support bath consists of a material such as closely packed gelatin microparticles that acts as a Bingham plastic and maintains the intended shape and structure of the bioink after printing. Once the printed construct has solidified, the support bath can then be dissolved away by increasing the temperature to 37 °C7.
A recent review article summarized the materials that have been 3D bioprinted in various publications using FRESH technique. These naturally derived materials range from collagen type I to methacrylated hyaluronic acid and represent several different gelation mechanisms7. Most research studies performed using this 3D bioprinting technique employ static biomaterials that do not change in response to external stimuli. Dynamic phototunable hydrogel biomaterials have been used by our lab and others8,9,10,11,12 to model a variety of fibrotic diseases. Unlike static biomaterials, phototunable bioinks allow for a softened model with lower elastic modulus value to be created and later stiffened to explore cellular responses to increases in microenvironmental stiffening.
Fibrotic diseases are characterized by an increase in the extracellular matrix production that can cause scarring and stiffening13. Tissue stiffening can initiate further injury and destruction of the impacted tissue, causing permanent organ damage and even death; fibrotic disorders are responsible for one-third of mortality worldwide. Fibroblasts produce excess and aberrant extracellular matrix in this disease state14,15. Increased fibroblast proliferation and extracellular matrix deposition further stiffen the tissue and activates a profibrotic positive feedback loop16,17,18,19. Studying fibroblast activation is vital to understanding fibrotic diseases. Here we present human pulmonary arterial hypertension (PAH) as an example of one fibrotic disorder in which it is important to mimic the 3D geometry of the blood vessel using 3D bioprinting and introduce the dynamic stiffening capabilities of phototunable hydrogels. PAH is a condition in which pressure in the main pulmonary arteries surpasses normal levels and applies strain to the heart, increasing human pulmonary artery adventitial fibroblast (HPAAF) activation and stiffens the blood vessel tissues16,17,18,19. A phototunable poly(ethylene glycol)-alpha methacrylate (PEGαMA) bioink formulation allows for temporal stiffening in constructs and helps model both healthy tissue and disease progression5,8,9,10. Exploiting this unique feature enables the quantification of HPAAF activation and proliferation in response to microenvironmental stiffening in 3D and may provide valuable insight into the cellular mechanisms involved in this disease. The protocol described here will allow researchers to create 3D models that recapitulate changes in the extracellular microenvironment during disease progression or tissue repair and study fibroblast activation.
Protocol
1. PEGαMA synthesis and characterization
NOTE: Poly(ethylene glycol)-alpha methacrylate (PEGαMA) synthesis was adapted from Hewawasam et al. and performed under moisture-free conditions9.
- Weigh the reactants.
NOTE: For example, weigh out 5 g 10 kg/mol 8-arm PEG-hydroxyl (PEG-OH) and 0.38 g sodium hydride (NaH) (see Table of Materials). - Add a stir bar to 250 mL Schlenk flask and purge with argon.
- Dissolve the PEG-OH in the lowest volume of anhydrous tetrahydrofuran (THF) required for dissolution within the Schlenk flask.
NOTE: Approximately 80 mL of THF will dissolve 5 g PEG-OH. Add the minimal amount of THF required to dissolve the PEG-OH. - Add 3 times molar excess NaH to the reaction mixture and stir at room temperature (RT) for 30 min.
- Add 6 times molar excess Ethyl 2-(bromomethyl)acrylate (EBrMA, see Table of Materials) dropwise to the Schlenk flask and cover the reaction vessel with aluminum foil to protect it from light. Stir the reaction at room temperature for approximately 48 h.
NOTE: For 5 g PEG-OH and 0.38 g NaH, use 3.68 mL EBrMA for this reaction. - Add a few drops of 1 N acetic acid to quench the reaction. Vacuum filter the solution through a filtration aid.
NOTE: Adding acetic acid will produce gas bubbles. Stop adding acetic acid drops when bubbles stop forming as this indicates the mixture has been quenched successfully. - Concentrate the filtrate on a rotary evaporator and precipitate in 4 °C diethyl ether. Leave precipitate protected from light at 4 °C for 12-18 h.
- Add a Whatman filter paper to a Buchner funnel. Slowly pour the reaction mixture over the filter paper and use vacuum suction to separate the precipitate from diethyl ether. Collect the precipitate in a dry and clean filtration flask.
- Vacuum dry the product for at least 5 h or overnight at room temperature and dissolve in minimum volume of deionized water needed. Transfer the dissolved product into dialysis tubing (see Table of Materials) and dialyze against 3.5 L of deionized water for at least four days. Change the dialysis water every 12 h.
NOTE: The product will appear as completely dry pure white solid powder after vacuum drying. - Flash-freeze the product and lyophilize for approximately 72 h or until it is completely dry.
- Dissolve the product in chloroform D (CDCl3). Run the sample using 1H NMR with a protocol that performs 248 scans with 2.5 s relaxation time.
- Verify the functionalization and purity of the product by calibrating CDCl3 solvent peak to 7.26 PPM. Integrate the peak for PEG backbone protons (d3.71) and calibrate the integration to 114.
- Integrate the remaining peaks: PEGαMA 1H NMR (300 MHz, CDCl3): d (ppm) 1.36 (t, 3H, CH3-), 3.71 (s, 114H, PEG CH2-CH2), 4.29 (t, s, 4H, -CH2-C(O)-O-O, -O-CH2-C(=CH2)-), 5.93 (q, 1H, -C=CH2), 6.34 (q, 1H, -C=CH2) and compare the integration for the αMA alkene end group peaks to the expected value (1H) based on the PEG backbone calibration (Figure 1).
NOTE: Average the two peaks labeled as "a" (Figure 1) and multiply by 100 to obtain the average PEGαMA functionalization percentage.
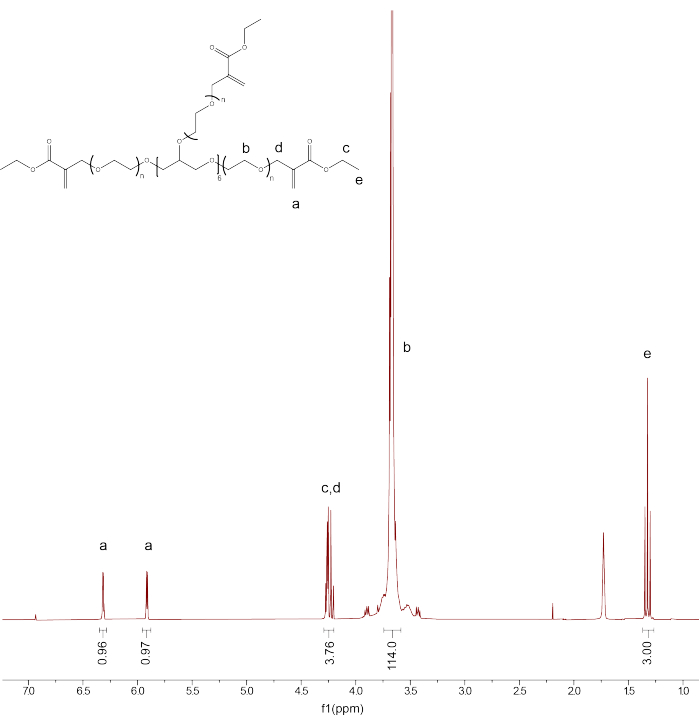
Figure 1: Proton NMR confirmed successful PEGαMA functionalization. NMR analysis was performed in chloroform-D (CDCl3) and showed the functionalization of 96.5%. PEGαMA 1H NMR (300 MHz, CDCl3): d (ppm) 1.36 (t, 3H, CH3-), 3.71 (s, 114H, PEG CH2-CH2), 4.29 (t, s, 4H, -CH2-C(O)-O-O, -O-CH2-C(=CH2)-), 5.93 (q, 1H, -C=CH2), 6.34 (q, 1H, -C=CH2). Please click here to view a larger version of this figure.
2. Model design and 3D bioprinter setup
NOTE: A commercially available 3D printer (see Table of Materials) was modified by replacing the thermoplastic extruder with a custom-built syringe pump extruder and adapted from Hinton et al.20. Open-source designs are available online: https://3d.nih.gov/users/awfeinberg.
- Open Fusion 360 software (see Table of Materials) and make a 3D computer-aided hollow cylinder design.
NOTE: A downloadable file that can be used for this step and mimics blood vessel geometry can be found in Supplementary File 1. - Save the file and open it within the Slic3r software (see Table of Materials). Double check that all the parameters are as desired, and then push the export G-code button. Save the G-code to the computer.
- Open the Pronterface software (see Table of Materials) and upload the G-code file.
NOTE: The Pronterface software interfaces with the bioprinter and provides sufficient hardware input control. A usable G-code file can be found in Supplementary File 2. - Transfer the bioprinter and all associated parts into a biosafety cabinet (BSC) using aseptic techniques.
- Assemble a 30 G 0.5" length blunt needle tip (see Table of Materials) to the printing glass syringe and set aside.
- Plug the power cord of the bioprinter into an outlet. Press the red power button on the front of the bioprinter to turn it on. Connect the universal serial bus (USB) cord between the computer and bioprinter and ensure all wire connections are established and plugged in.
3. Preparation of the support bath and reagents
NOTE: Perform all steps in a biosafety cabinet using aseptic techniques.
- Prepare cell culture medium consisting of SmBM Basal Medium (CC-3181) and SmGM-2 SingleQuots supplements (CC-4149), excluding fetal bovine serum (FBS), according to the manufacturer's instructions. Store at 4 °C until use.
- Aliquot 50 mL of the cell culture medium and add 1% v/v of FBS (CC-4149) (see Table of Materials) to make a low-serum media. Store at 4 °C until use.
- Resuspend the gelatin slurry powder according to the manufacturer's instructions using sterile cell culture media without FBS as the solvent (see Table of Materials). Immediately before use, adjust the final pH of the the gelatin slurry to pH 9 using 2 M potassium hydroxide (KOH) and/or 2 M hydrochloric acid (HCl) to adjust the solution pH as needed using a pH meter.
- Fill the desired number of wells of a 24-well plate each approximately half full using 1 mL of gelatin slurry per well using a syringe without the needle.
NOTE: Uniformly fill the center of the wells and confirm that no air pockets exist. Tap the plate to help evenly distribute the microparticle slurry. Adjust the height and volume of slurry per well as needed to accommodate each bioprint size and shape. Users can create a homemade syringe to transfer the gelatin slurry into each well. This can be done by adding a correctly sized syringe plunger into a 50 mL test tube already containing the compacted gelatin slurry at the bottom. While inserting the plunger, insert a small guide wire alongside the test tube to help air escape, and then remove it when the plunger is in contact with the gelatin slurry. Immediately before use, cut off the tip of the test tube with a razor blade to create a hole for the gelatin slurry to extrude out of and press down on the plunger. - Place the filled 24-well plate onto the center of the bioprinter stage and secure it to the stage.
NOTE: Figure 2 shows a generic bioprinter setup. Place a rubber band around the print stage to secure a 24-well plate to the platform and prevent movement.

Figure 2: Basic 3D bioprinting setup. The bioprinter was set up within a sterile environment such as a biosafety cabinet, and the printhead was assembled so that the glass syringe and needle were vertically lowered into the support bath printing area below. Please click here to view a larger version of this figure.
4. Cell culture
NOTE: Perform all steps in a biosafety cabinet using aseptic techniques.
- Thaw HPAAF cells (commercially obtained, see Table of Materials) and expand them out in T-75 tissue culture-treated plastic flasks containing SmBM Basal Medium (CC-3181) and all the SmGM-2 SingleQuots supplements (CC-4149) according to the manufacturer's instructions (see Table of Materials).
NOTE: Standard cell culture protocols for adherent cells should be used, maintaining the cells at 37 °C and 5% CO2 and replenishing the media every few days. - Once the HPAAFs have reached ~80-90% confluency, aspirate the media and rinse the cells one time with phosphate-buffered saline (PBS).
- Add approximately 4 mL of prewarmed 0.05% trypsin-EDTA to each T-75 flask. Tilt the flask to ensure that the entire cell culture surface is covered with 0.05% Trypsin-EDTA solution. Incubate the flasks for 3-5 min at 37 °C and check for cell detachment.
- Once the cells are floating, add at least 6 mL of Dulbecco's Modified Eagle Medium (DMEM) to each flask and transfer the cells into a 50 mL conical tube.
- Centrifuge the cell suspension at 300 x g for 5 min at room temperature to pellet the cells. Aspirate the supernatant from the cell pellet and resuspend the cells in 1-3 mL media with FBS using a 1000 µL pipette, ensuring a single-cell suspension.
- Transfer 10 µL of the cell suspension to a microcentrifuge tube. Add 10 µL of Trypan Blue solution and mix well. Use 10 µL of this mixture to count cells within a hemocytometer using an inverted light microscope.
NOTE: To achieve a 4 x 106 cells/mL final bioink concentration, 800,000 fibroblasts were set aside for each 200 µL of bioink.
5. Preparation of hydrogel bioink
NOTE: Bioink preparation was adapted from Davis-Hall et al.5. Steps 5.1-5.2 can be completed in parallel with steps 4.1-4.3 to minimize the time between cell collection and resuspension in the bioink. Perform steps in a biosafety cabinet using an aseptic technique.
- Prepare a 20 mM tris(2-carboxyethyl) phosphine (TCEP, see Table of Materials) pH 7 solution and sterile filter using a 0.2 µm syringe filter. Immediately before use, add 2 M KOH and/or 2 M HCl to adjust the solution pH as needed. Measure with pH meter and adjust accordingly.
NOTE: TCEP reduces disulfide bonds. - Prepare a 0.25 mg/mL stock solution of PEGαMA resuspended in sterile cell culture media without FBS, 250 mM stock solutions of 1,4-dithiothreitol (DTT), MMP2-degradable crosslinker, and CGRGDS (RGD) peptide (see Table of Materials), resuspending all in 20 mM sterile TCEP, and a 15 wt% stock solution of poly(ethylene oxide) (PEO) in distilled (DI) water using pipettes as needed.
- Following Table 1 as a guide, combine the respective amounts needed of PEGαMA, DTT, MMP2-degradable crosslinker, PEO, CGRGDS, and low-serum cell culture media together with the fibroblasts in a 50 mL conical tube.
NOTE: It is recommended to check the pH with pH strips after adding all but the cell culture media as this combination should result in the pH being very close to 6.2. If further pH adjustments are required, keep track of how much additional volume is needed to adjust the pH of the precursor solution. Bring up the total volume to 200 µL by adding the remaining cell culture media volume minus any volume added during the final pH adjustment. - Mix the bioink together using a positive displacement pipette to ensure the cells are single cells and confirm the final precursor solution is pH 6.2 to prevent base-catalyzed polymerization during 3D bioprinting.
- Load the bioink into the glass syringe by removing the plunger and using a separate syringe with a 15 gauge 1.5" length blunt needle tip (see Table of Materials) attached to transfer the bioink from the centrifuge tube to the syringe, being careful to avoid forming air bubbles within the solution.
- Place the glass syringe within the print head and attach print head components so that everything is firmly assembled and ready for printing.
NOTE: At this point, the glass syringe within the print head should have a 30 gauge 0.5" length blunt needle tip attached to it for printing.
| Component | Stock Solution Concentration | Amount to Add |
| PEGαMA | 0.25 mg/ml | 140 µL |
| DTT | 250 mM | 12.24 µL |
| MMP2 Degradable Crosslinker | 250 mM | 5.25 µL |
| RGD | 250 mM | 1.6 µL |
| PEO | 15 wt% | 33.33 µL |
| Activation Media and/or pH Adjustment Reagents | - | 7.58 µL |
| Fibroblasts | - | 800000 cells |
Table 1: Example volumes required to prepare 200 µL of bioink (hydrogel precursor solution and fibroblast cells).
6. 3D bioprinting
NOTE: Perform all steps in a biosafety cabinet using aseptic techniques.
- Using the directional arrows within the Pronterface software, manually adjust the position of the extrusion needle into the center of a well and within the support bath slurry. Leave at least 1 mm support bath slurry below the tip of the needle.
NOTE: The software has no ability to tell where the needle is within space. It is completely up to the user to move the needle by manually clicking the arrows within the software (e.g., clicking the up arrow will move the needle up or away from the printing platform, etc.). Maneuver the needle carefully to ensure it will not hit any boundaries of the well. - Once the needle tip is situated in the center of the slurry within the well, hit the start button within the Pronterface and wait for the print to complete to achieve constructs, as demonstrated in Figure 3A.
NOTE: To bioprint one construct using the provided file (Supplementary File 1), it will take approximately 3 min. It takes approximately 5 min to orient and move the needle and then print one construct completely from start to finish. - Repeat steps 6.1-6.2 until the number of desired bioprinted constructs is met.
NOTE: It is recommended to make more constructs than needed to account for any unsuccessful prints. If failure occurs, move to the next well, reset everything, and repeat steps 6.1-6.2 again. - Leave the well-plate at room temperature and cover it in the BSC for 1 h after the printing has finished to allow for base-catalyzed polymerization of the phototunable hydrogel.
- Place the well plate containing 3D bioprinted constructs into a 37 °C sterile incubator and leave them for 12-18 h to melt away the support bath slurry.
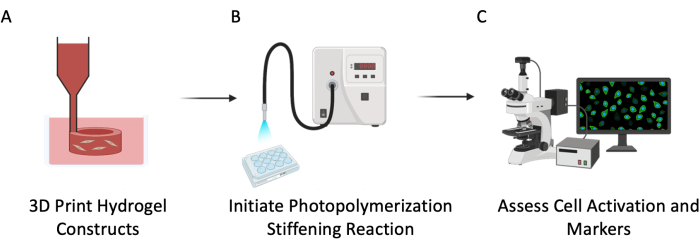
Figure 3: Experimental schematic. This protocol was described in three major steps: (A) 3D bioprinting PEGαMA hollow tubes with embedded cells to mimic pulmonary vasculature. (B) Photoinitiation of homopolymerization reaction to stiffen the cellular microenvironment. (C) Assessment of cellular markers for proliferation and activation. Please click here to view a larger version of this figure.
7. 3D bioprinted construct culture and photostiffening
NOTE: All steps should be performed in a biosafety cabinet using aseptic techniques.
- Prepare 2.2 mM lithium phenyl-2,4,6-trimethylbenzoylphosphinate (LAP) (see Table of Materials) stock solution in PBS and sterile filter using a 0.2 µm syringe filter. Keep the LAP solution protected from light.
- After 12-18 h, change the media surrounding the bioprinted constructs. Manually remove the media and melted gelatin support bath within the wells and be careful not to disturb the bioprinted constructs.
NOTE: It is helpful to slowly remove the media while holding the plate at a 45° angle so that the constructs emerge within the well and can be seen. A clear hydrogel cylinder should be identifiable in each well with successful print (Figure 4A). - Add an appropriate volume of low-serum media to each well.
NOTE: For a 24-well plate, 700 µL media per well should completely cover the bioprinted constructs. Adjust as needed. - Return the plate to the incubator and change the media on the samples every 3 days or in accordance with the experimental design.
- Twenty-four hours before the desired stiffening time point, remove media from the samples and replace them with low-serum media supplemented with 2.2 mM sterile LAP.
NOTE: To fluorescently label the structure, swell 3D bioprinted constructs in PBS supplemented with 10 µM methacryloxyethyl thiocarbamoyl rhodamine B (see Table of Materials) overnight, then stiffen as described in step 7.6 to fluorescently label the structure. Transfer into PBS for 2 days at 4 °C to remove excess rhodamine and image using a TRITC filter (Figure 4B,C). - At the desired stiffening time point, remove half of the media from the wells to be stiffened and place the plate with the lid off under UV light. Turn on the UV light and stiffen these constructs by applying 10 mW/cm2 365 nm light for 5 min using Omnicure (see Table of Materials) and a 365 nm bandpass filter (Figure 3B).
NOTE: Use the radiometer/photometer to confirm the light intensity is correct before exposing cells to UV light. - Remove the remaining media from these wells and add fresh low-serum media to each well. Return the plate to the incubator.
- Take out the plate from the incubator and perform the fibroblast activation study at the desired time point following step 9.
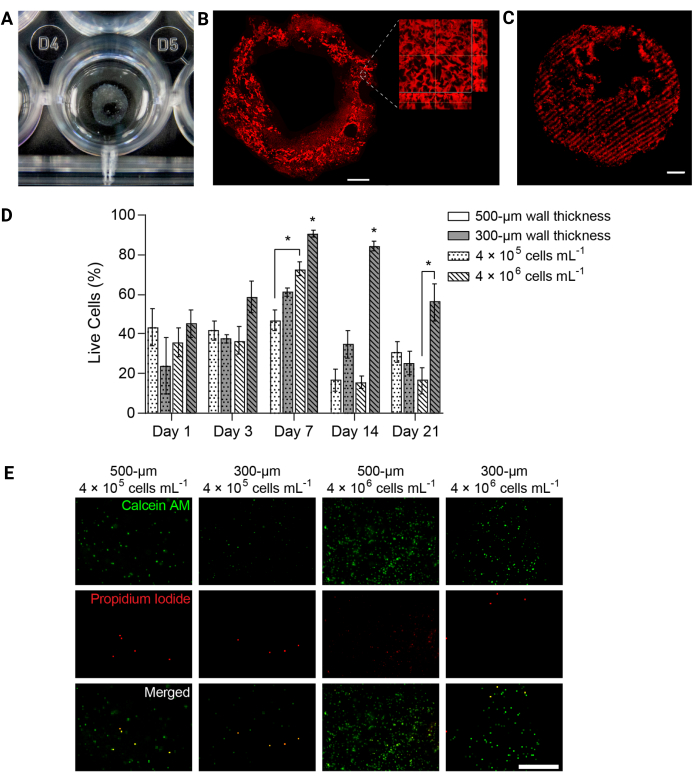
Figure 4: 3D-bioprinted hydrogel structures supported cell viability over time. (A) Photograph of 3D-printed hydrogel structure in a 24-well plate. (B) Maximum intensity projection of fluorescently labeled PEGαMA 3D-printed hydrogel. Scale bar = 1 mm. Higher magnification microscopy showed pores within the hydrogel structure induced by gelatin microparticles in the FRESH bioprinting support bath. (C) 3D-printed PEGαMA tube with fluorescently labeled stiffened regions imaged on a confocal microscope (100 µm z-stack displayed as a maximum intensity projection) showed spatial control over stiffening in 3D. Scale bar = 500 µm. (D) HPAAF viability in 3D-bioprinted constructs measured by Live/Dead assays. Constructs with 300 µm thickness and 4 × 106 cells/mL outperformed all other conditions at every time point. Viability peaked on day 7. This condition and time point were selected for future experiments. Columns show mean ± SEM, n = 3. *, p < 0.05, ANOVA, Tukey HSD. (E) Representative confocal images of cells in 3D constructs stained with live/dead reagent at day 7, the time point with the greatest overall viability. Calcein AM marked live cells in green and propidium iodide marked dead cells in red. The right-most column shows that the best-performing condition had a uniform cell distribution and a high percentage of live cells. Scale bar = 500 µm. Reproduced with permission from Davis-Hall et al.5. Please click here to view a larger version of this figure.
8. The assessment of fibroblast viability
- At desired viability time points, stain using calcein AM and propidium iodide (see Table of Materials).
NOTE: To summarize, media should be removed from each well and the constructs should be rinsed with sterile PBS. Incubate the constructs in a live/dead staining solution for 40 min at 37°C on a rocker. The staining solution should contain calcein AM (1:1000 dilution) and propidium iodide (1:1000 dilution) to identify live or dead cells upon imaging. - Transfer the constructs into sterile PBS and immediately image on a confocal fluorescence microscope. Acquire three different 100 µm z-stack images per sample per time point and express viability as the average percentage of live cells (Figure 4D,E).
9. The assessment of fibroblast activation
- Prepare 3% w/v bovine serum albumin (BSA) and 0.1% v/v Tween 20 in PBS. This solution will be referred to as the immunofluorescence (IF) solution.
- At desired timepoints, remove media from the sample wells and rinse the constructs with PBS. Replace the PBS with 4% paraformaldehyde (PFA) and keep these samples at 37 °C for 30 min on a rocker. Then, replace the 4% PFA with 100 mM glycine in PBS and leave these samples on a rocker at room temperature (RT) for another 15 min.
- Next, transfer these samples into Tissue-Tek Cryomolds, cover the sample completely with optimal cutting temperature (OCT) solution (see Table of Materials), and allow the OCT to diffuse into the samples for 12-18 h at 4 °C.
- Flash-freeze the OCT-soaked samples in 2-methylbutane using liquid nitrogen. Fill a styrofoam box or other appropriate container with liquid nitrogen and then place a second container filled with 2-methylbutane within the liquid nitrogen so that it is at least halfway submerged. Use forceps to hold each cryomold containing an OCT-covered sample in liquid nitrogen cooled 2-methylbutane until visibly frozen. These samples can be stored at -80 °C until they are ready for cryosection.
CAUTION: Personal protective equipment (PPE) such as cold protectant gloves, a cold protectant apron, and face shield provided in the Cryogenic Safety Kit (see Table of Materials) should be used while handling liquid nitrogen. - Cryosection the frozen OCT samples at -22 °C and attach 10 µm thick slices to positively charged glass microscope slides. Prepare three microscope slides with at least 3-5 cryosections per slide for each 3D hydrogel sample.
NOTE: The microscope slides can be stored at this point at -80 °C if needed as a stopping point. - Fix the cryosections in ice-cold acetone for 15 min to help the cryosections adhere to the slides. Gently rinse the cryosections with RT water to remove any remaining OCT. Let these samples dry and outline the cryosections with a hydrophobic pen (see Table of Materials).
- Permeabilize the samples at room temperature with 0.2% v/v Triton X-100 in PBS for 10 min and then block the sections with 5% BSA w/v in PBS for 1 h at RT.
- Add the primary mouse anti-human alpha smooth muscle actin (αSMA) antibody (1:250 dilution) (see Table of Materials) to the IF solution. Store these sectioned samples with the primary antibody on them overnight at 4 °C. Rinse the samples 3x with IF solution.
- Incubate the sections in IF solution containing the secondary goat anti-mouse Alexa Fluor 555 antibody (1:250 dilution) and two drops of ActinGreen 488 ReadyProbe (see Table of Materials) per milliliter of IF solution. Cover the samples for all subsequent steps with aluminum foil to protect them from light and let the secondary antibody solution stay on the samples for 1 h at RT.
- Rinse the sections 3x with IF solution. Incubate them in 300 nM 4',6-diamidino-2-phylindole (DAPI) in DI water for 15 min at RT. Conduct a final rinse of the sections 3x with DI water.
- Using 10 µL of a commercially available antifade reagent (see Table of Materials), coverslip the sections using standard methods.
NOTE: The mounted slides can be stored protected from light in a -80 °C freezer until needed for imaging. - Image the cryosections using a fluorescence microscope (Figure 3C and Figure 5B). Image three random sections per slide using a 10x objective.
NOTE: Images should be taken in the DAPI, FITC, and TRITC channels. - Upload the images into ImageJ (NIH). Quantify the percentage of αSMA positive cells as a measurement of fibroblast activation (Figure 5A) by dividing the total number of αSMA-positive cells by the total number of cell nuclei for each field of view.
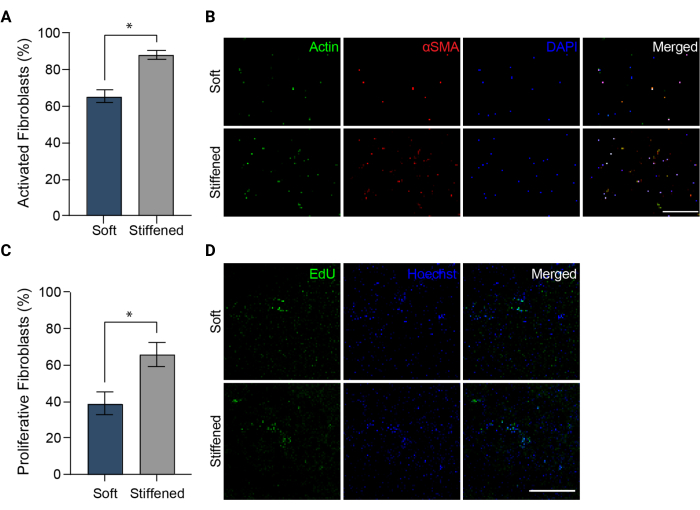
Figure 5: Fibroblast activation in 3D-bioprinted models of pulmonary arterial adventitia. (A) Fibrotic activation in soft and stiffened 3D hydrogels measured by αSMA expression. HPAAFs in stiffened constructs were significantly more positive for αSMA than cells in soft constructs. Columns represent mean ± SEM, n = 3. *, p < 0.05, Mann-Whitney U test. (B) Representative confocal images of immunostaining for αSMA, actin, and DAPI in soft and stiffened 3D hydrogels. HPAAFs in stiffened constructs showed more prevalent αSMA immunofluorescence than cells in soft constructs. Scale bar = 250 µm. (C) Fibroblast proliferation in soft and stiffened 3D bioprinted constructs measured by EdU positivity. HPAAFs in stiffened constructs were significantly more positive for EdU than cells in soft constructs. Columns represent mean ± SEM, n = 3. *, p < 0.05, Mann-Whitney U test. (D) Representative confocal images of immunostaining for EdU and Hoechst dye in soft and stiffened 3D hydrogels. HPAAFs in stiffened constructs showed more prevalent EdU immunofluorescence than cells in soft constructs. Scale bar = 300 µm. Reproduced with permission from Davis-Hall et al.5. Please click here to view a larger version of this figure.
10. The assessment of fibroblast proliferation
- Twenty-four hours before a desired proliferation time point, remove cell culture media from each well and replace with low-serum media supplemented with 10 µM EdU solution from the commercially available cell proliferation kit (see Table of Materials). Return the samples to the incubator for overnight incubation.
- At the desired proliferation time point, fix the samples incubated with EdU using 4% PFA at 37 °C for 30 min on a rocker. Replace the 4% PFA solution with 100 mM glycine in PBS and incubate samples at 37 °C for at least 15 minutes. Add the Hoechst at an appropriate concentration for 30 min and then rinse the constructs 2x with PBS.
NOTE: The samples can be stored protected from light at 4 °C until imaging. - Image all fixed and stored EdU samples using a fluorescence microscope and suggested cell proliferation kit manufacturer settings and filters (Figure 3C and Figure 5D). Acquire three different 100 µm z-stack images per sample and create maximum projections from each of these z-stacks. Measure HPAAF proliferation by counting the number of EdU-positive cells and dividing by the total number of cells identified by Hoechst counterstain within the maximum projection images (Figure 5C).
Results
This protocol describes how to 3D bioprint phototunable hydrogels within a support bath to create constructs capable of dynamic and temporal stiffening for studying fibroblast activation in geometries that mimic human tissues. First, the protocol explained how to synthesize PEGαMA, the backbone of this phototunable polymer system. Nuclear magnetic resonance (NMR) spectroscopy measurements showed successful PEGαMA functionalization at 96.5% (Figure 1). Functionalization values of 90...
Discussion
Dual-stage polymerization reactions in response to controlled light exposure can stiffen biomaterials with spatial and temporal control. Several studies have harnessed this technique to evaluate cell-matrix interactions in various platforms5,8,9,10,11,21,22,23
Disclosures
The authors do not have any conflicts of interest to disclose. Portions of this manuscript are reproduced with permission from © IOP Publishing https://doi.org/10.1088/1758-5090/aca8cf.5 All rights reserved.
Acknowledgements
The authors would like to acknowledge Dr. Adam Feinberg (Carnegie Mellon University) and those who hosted the 3D Bioprinting Open-Source Workshop. These individuals made it possible to learn the techniques of FRESH bioprinting and build the 3D bioprinter used for these studies. Additionally, the authors would like to acknowledge Biorender.com, which was used to produce figures in this manuscript. This work was supported by multiple groups or funding sources including the Rose Community Foundation (DDH and CMM), a Colorado Pulmonary Vascular Disease Research Award (DDH and CMM), the National Science Foundation under Award 1941401 (CMM), the Department of the Army under Award W81XWH-20-1-0037 (CMM), the National Cancer Institute of the NIH under Award R21 CA252172 (CMM), the Ludeman Family Center for Women's Health Research at the University of Colorado Anschutz Medical Campus (DDH and CMM), the National Heart, Lung, and Blood Institute of the National Institutes of Health under Awards R01 HL080396 (CMM), R01 HL153096 (CMM), F31 HL151122 (DDH), and T32 HL072738 (DDH and AT).
Materials
| Name | Company | Catalog Number | Comments |
| AccuMax Radiometer/Photometer Kit | Spectronics Corporation | XPR-3000 | To measure light intensity, used for photostiffening |
| Acetic Acid | Fisher Scientific | BP2401-500 | Used during PEGaMA synthesis |
| Acetone | Fisher Scientific | A184 | Used with the cryosections |
| ActinGreen 488 ReadyProbes | Fisher Scientific | R37110 | Used for staining |
| Aluminum Foil | Reynolds | F28028 | |
| Anhydrous Tetrahydrofuran (THF) | Sigma-Aldrich | 401757-1L | Used during PEGaMA synthesis |
| Argon Compressed Gas | Airgas | AR R300 | Used during PEGaMA synthesis |
| 8 Arm Poly(ethylene glycol)-hydroxyl (PEG-OH) | JenKem Technology | 8ARM-PEG-10K | Used during PEGaMA synthesis |
| 365 nm Bandpass Filter | Edmund Optics | 65-191 | Used for photostiffening |
| Bovine Serum Albumin (BSA) | Fisher Scientific | BP9700-100 | Used during staining process |
| Buchner Funnel | Quark Glass | QFN-8-14 | Used during PEGaMA synthesis |
| Calcein AM | Invitrogen | 65-0853-39 | Used during staining process |
| Celite 545 (Filtration Aid) | EMD Millipore | CX0574-1 | Used during PEGaMA synthesis |
| Charged Microscope Slides | Globe Scientific | 1358W | |
| Chloroform-d | Sigma-Aldrich | 151823-10X0.75ML | Used to characterize PEGaMA |
| Click-iT Plus EdU Cell Proliferation Kit | Invitrogen | C10637 | Used for staining |
| 50 mL Conical Tubes | CELLTREAT | 667050B | |
| Cryogenic Safety Kit | Cole-Parmer | EW-25000-85 | |
| Cryostat | Leica | CM 1850-3-1 | |
| Dialysis Tubing | Repligen | 132105 | |
| 4’,6-Diamidino-2-Phylindole (DAPI) | Sigma-Aldrich | D9542-1MG | Used for staining |
| Diethyl Ether | Fisher Scientific | E1384 | Used during PEGaMA synthesis |
| 1,4-Dithiothreitol (DTT) | Sigma-Aldrich | 10197777001 | Bioink component |
| Dulbecco's Modified Eagle's Medium (DMEM) | Cytiva | SH30271.FS | |
| Ethyl 2-(Bromomethyl)Acrylate (EBrMA) | Ambeed Inc. | A918087-25g | Used during PEGaMA synthesis |
| Filter Paper | Whatman | 1001-090 | Used during PEGaMA synthesis |
| Freezone 2.5L Freeze Dry System | Labconco | LA-2.5LR | Lyophilizer |
| Fusion 360 | Autodesk | N/A | Software download |
| 2.5 mL Gastight Syringe | Hamilton | 81420 | Used for bioprinting |
| 15 Gauge 1.5" IT Series Tip | Jensen Global | JG15-1.5X | Used for bioprinting |
| 30 Gauge 0.5" HP Series Tip | Jensen Global | JG30-0.5HPX | Used for bioprinting |
| Goat Anti-Mouse Alexa Fluor 555 Antibody | Fisher Scientific | A21422 | Used for staining |
| Glycine | Fisher Scientific | C2H5NO2 | Used during staining process |
| Hemocytometer | Fisher Scientific | 1461 | |
| Hoechst | Thermo Scientific | 62249 | Used during staining process |
| Human Pulmonary Artery Adventitial Fibroblasts (HPAAFs) | AcceGen | ABC-TC3773 | From a 2-year-old male patient |
| Hydrochloric Acid (HCl) | Fisher Scientific | A144-500 | Used to pH adjust solutions |
| ImageJ | National Institutes of Health (NIH) | N/A | Free software download |
| ImmEdge® Pen | Vector Laboratories | H-4000 | Used during staining process |
| Incubator | VWR | VWR51014991 | |
| LifeSupport Gelatin Microparticle Slurry (Gelatin Slurry) | Advanced Biomatrix | 5244-10GM | Used for bioprinting |
| Light Microscope | Olympus | CKX53 | Inverted light microscope |
| Lithium Phenyl-2,4,6-Trimethylbenzoylphosphinate (LAP) | Sigma-Aldrich | 900889-5G | Photoinitiator used for photostiffening |
| Liquid Nitrogen | N/A | N/A | |
| LulzBot Mini 2 | LulzBot | N/A | Bioprinter adapted |
| Methacryloxyethyl Thiocarbamoyl Rhodamine B | Polysciences Inc. | 669775-30-8 | |
| 2-Methylbutane | Sigma-Aldrich | M32631-4L | |
| Microman Capillary Pistons CP1000 | VWR | 76178-166 | Positive displacement pipette tips |
| MMP2 Degradable Crosslinker (KCGGPQGIWGQGCK) | GL Biochem | N/A | Bioink component |
| Mouse Anti-Human αSMA Monoclonal Antibody | Fisher Scientific | MA5-11547 | Used for staining |
| OmniCure Series 2000 | Lumen Dynamics | S2000-XLA | UV light source used for photostiffening |
| Paraformaldehyde (PFA) | Electron Microscopy Sciences | 15710 | Used to fix samples |
| pH Meter | Mettler Toledo | FP20 | |
| pH Strips | Cytiva | 10362010 | |
| Phosphate Buffered Saline (PBS) | Hyclone Laboratories, Inc. | Cytiva SH30256.FS | |
| Pipette Set | Fisher Scientific | 14-388-100 | |
| 10 µL Pipette Tips | USA Scientific | 1120-3710 | |
| 20 µL Pipette Tips | USA Scientific | 1183-1510 | |
| 200 µL Pipette Tips | USA Scientific | 1111-0700 | |
| 1000 µL Pipette Tips | USA Scientific | 1111-2721 | |
| Poly(Ethylene Glycol)-Alpha Methacrylate (PEGαMA) | N/A | N/A | Refer to manuscript for synthesis steps |
| Poly(Ethylene Oxide) (PEO) | Sigma-Aldrich | 372773-250G | Bioink component |
| Positive Displacement Pipette | Fisher Scientific | FD10004G | 100-1000 µL |
| Potassium Hydroxide (KOH) | Sigma-Aldrich | 221473-500G | Used to pH adjust solutions |
| ProLong Gold Antifade Reagent | Invitrogen | P36930 | Used during staining process |
| Pronterface | All3DP | N/A | Software download |
| Propidium Iodide | Sigma-Aldrich | P4864-10ML | Used for staining |
| RGD Peptide (CGRGDS) | GL Biochem | N/A | Bioink component |
| Rocker | VWR | 10127-876 | |
| Rotary Evaporator | Thomas Scientific | 11100V2022 | Used during PEGaMA synthesis |
| Rubber Band | Staples | 808659 | |
| Schlenk Flask | Kemtech America | F902450 | Used during PEGaMA synthesis |
| Slic3r | Slic3r | N/A | Software download |
| Smooth Muscle Cell Growth Medium-2 (SmGM-2) BulletKit | Lonza | CC-3182 | Kit contains CC-3181 and CC-4149 components |
| Sodium Hydride | Sigma-Aldrich | 223441-50G | Used during PEGaMA synthesis |
| Sorvall ST 40R Centrifuge | Fisher Scientific | 75-004-525 | |
| Stir Bar | VWR | 58948-091 | |
| Syringe Filter | VWR | 28145-483 | Used to sterile filter solutions |
| T-75 Tissue-Cultured Treated Flask | VWR | 82050-856 | Used for cell culture work |
| Tissue-Tek Cyromold | Sakura | 4557 | |
| Tissue-Tek O.C.T Compound (OCT) | Sakura | 4583 | |
| Tris(2-Carboxyethyl) Phosphine (TCEP) | Sigma-Aldrich | C4706-2G | |
| Triton X-100 | Fisher Bioreagents | C34H622O11 | Used during staining process |
| Trypan Blue | Sigma-Aldrich | T8154-20ML | Used for cell culture work |
| 0.05% Trypsin-EDTA | Gibco | 25-300-062 | Used for cell culture work |
| Tween 20 | Fisher Bioreagents | C58H114O26 | Used during staining process |
| Upright Microscope | Olympus | BX63F | Fluorescent microscope capabilities |
| Water Bath | PolyScience | WBE20A11B | |
| 24-Well Tissue Culture Plates | Corning | 3527 |
References
- Ahrens, J. H., et al. Programming cellular alignment in engineered cardiac tissue via bioprinting anisotropic organ building blocks. Advanced Materials. 34 (26), e2200217 (2022).
- Lin, N. Y. C., et al. Renal reabsorption in 3D vascularized proximal tubule models. Proceedings of the National Academy of Sciences of the United States of America. 116 (12), 5399-5404 (2019).
- Grigoryan, B., et al. Multivascular networks and functional intravascular topologies within biocompatible hydrogels. Science. 364 (6439), 458-464 (2019).
- Kang, Y., Datta, P., Shanmughapriya, S., Ozbolat, I. T. 3D bioprinting of tumor models for cancer research. ACS Applied Biomaterials. 3 (9), 5552-5573 (2020).
- Davis-Hall, D., Thomas, E., Pena, B., Magin, C. M. 3D-bioprinted, phototunable hydrogel models for studying adventitial fibroblast activation in pulmonary arterial hypertension. Biofabrication. 15 (1), (2022).
- Mirdamadi, E., Tashman, J. W., Shiwarski, D. J., Palchesko, R. N., Feinberg, A. W. FRESH 3D bioprinting of a full-size model of the human heart. ACS Biomaterials Science & Engineering. 6 (11), 6453-6459 (2020).
- Shiwarski, D. J., Hudson, A. R., Tashman, J. W., Feinberg, A. W. Emergence of FRESH 3D printing as a platform for advanced tissue biofabrication. APL Bioengineering. 5 (1), 010904 (2021).
- Petrou, C. L., et al. Clickable decellularized extracellular matrix as a new tool for building hybrid hydrogels to model chronic fibrotic diseases in vitro. Journal of Materials Chemistry B. 8 (31), 6814-6826 (2020).
- Hewawasam, R. S., Blomberg, R., Serbedzija, P., Magin, C. M. Chemical modification of human decellularized extracellular matrix for incorporation into phototunable hybrid hydrogel models of tissue fibrosis. ACS Applied Materials & Interfaces. 15 (12), 15071-15083 (2023).
- Saleh, K. S., et al. Engineering hybrid hydrogels comprised healthy or diseased decellularized extracellular matrix to study pulmonary fibrosis. Cellular and Molecular Bioengineering. 15 (5), 505-519 (2022).
- Guvendiren, M., Burdick, J. A. Stiffening hydrogels to probe short- and long-term cellular responses to dynamic mechanics. Nature Communications. 3, 792 (2012).
- Rosales, A. M., Vega, S. L., DelRio, F. W., Burdick, J. A., Anseth, K. S. Hydrogels with reversible mechanics to probe dynamic cell microenvironments. Angewandte Chemie International Edition English. 56 (40), 12132-12136 (2017).
- Wynn, T. A., Ramalingam, T. R. Mechanisms of fibrosis: therapeutic translation for fibrotic disease. Nature Medicine. 18 (7), 1028-1040 (2012).
- Huertas, A., Tu, L., Humbert, M., Guignabert, C. Chronic inflammation within the vascular wall in pulmonary arterial hypertension: more than a spectator. Cardiovascular Research. 116 (5), 885-893 (2020).
- Kendall, R. T., Feghali-Bostwick, C. A. Fibroblasts in fibrosis: novel roles and mediators. Frontiers in Pharmacology. 5, 123 (2014).
- Parker, M. W., et al. Fibrotic extracellular matrix activates a profibrotic positive feedback loop. The Journal of Clinical Investigation. 124 (4), 1622-1635 (2014).
- Habiel, D. M., Hogaboam, C. Heterogeneity in fibroblast proliferation and survival in idiopathic pulmonary fibrosis. Frontiers in Pharmacology. 5, 2 (2014).
- Hu, C. J., Zhang, H., Laux, A., Pullamsetti, S. S., Stenmark, K. R. Mechanisms contributing to persistently activated cell phenotypes in pulmonary hypertension. The Journal of Physiology. 597 (4), 1103-1119 (2019).
- Li, M., et al. Emergence of fibroblasts with a proinflammatory epigenetically altered phenotype in severe hypoxic pulmonary hypertension. The Journal of Immunology. 187 (5), 2711-2722 (2011).
- Hinton, T. J., et al. Three-dimensional printing of complex biological structures by freeform-reversible embedding of suspended hydrogels. Science Advances. 1 (9), e1500758 (2015).
- Brown, T. E., et al. Secondary photocrosslinking of click hydrogels to probe myoblast mechanotransduction in three dimensions. Journal of the American Chemical Society. 140 (37), 11585-11588 (2018).
- Ondeck, M. G., et al. Dynamically stiffened matrix promotes malignant transformation of mammary epithelial cells via collective mechanical signaling. Proceedings of the National Academy of Sciences of the United States of America. 116 (9), 3502-3507 (2019).
- Caliari, S. R., et al. Stiffening hydrogels for investigating the dynamics of hepatic stellate cell mechanotransduction during myofibroblast activation. Scientific Reports. 6, 21387 (2016).
- Liu, F., et al. Feedback amplification of fibrosis through matrix stiffening and COX-2 suppression. Journal of Cell Biology. 190 (4), 693-706 (2010).
- Tschumperlin, D. J., Ligresti, G., Hilscher, M. B., Shah, V. H. Mechanosensing and fibrosis. The Journal of Clinical Investigation. 128 (1), 74-84 (2018).
- Chelladurai, P., Seeger, W., Pullamsetti, S. S. Matrix metalloproteinases and their inhibitors in pulmonary hypertension. European Respiratory Journal. 40 (3), 766-782 (2012).
- Caracena, T., et al. Alveolar epithelial cells and microenvironmental stiffness synergistically drive fibroblast activation in three-dimensional hydrogel lung models. Biomaterials Science. 10 (24), 7133-7148 (2022).
- Ruskowitz, E. R., DeForest, C. A. Proteome-wide analysis of cellular response to ultraviolet light for biomaterial synthesis and modification. ACS Biomaterials Science & Engineering. 5 (5), 2111-2116 (2019).
- Kruse, C. R., et al. The effect of pH on cell viability, cell migration, cell proliferation, wound closure, and wound reepithelialization: In vitro and in vivo study. Wound Repair and Regeneration. 25 (2), 260-269 (2017).
- Filippi, M., et al. Perfusable biohybrid designs for bioprinted skeletal muscle tissue. Advanced Healthcare Materials. , e1500758 (2023).
- Matthiesen, I., et al. Astrocyte 3D culture and bioprinting using peptide-functionalized hyaluronan hydrogels. Science and Technology of Advanced Materials. 24 (1), 2165871 (2023).
- Xu, L., et al. Bioprinting a skin patch with dual-crosslinked gelatin (GelMA) and silk fibroin (SilMA): An approach to accelerating cutaneous wound healing. Materials Today Bio. 18, 100550 (2023).
- Bliley, J. M., Shiwarski, D. J., Feinberg, A. W. 3D-bioprinted human tissue and the path toward clinical translation. Science Translational Medicine. 14 (666), eabo7047 (2022).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved