A subscription to JoVE is required to view this content. Sign in or start your free trial.
An Intraperitoneal Injection Technique in Adult Zebrafish that Minimizes Body Damage and Associated Mortality
* These authors contributed equally
In This Article
Summary
A new intraperitoneal (IP) injection method in adult zebrafish is described. When handling toxic compounds such as doxorubicin, this procedure is more effective than the two previously reported IP methods. The technique is designed to be easily adopted by researchers with limited experience in the zebrafish model.
Abstract
The adult zebrafish (Danio rerio), which is genetically accessible, is being employed as a valuable vertebrate model to study human disorders such as cardiomyopathy. Intraperitoneal (IP) injection is an important method that delivers compounds to the body for either testing therapeutic effects or generating disease models such as doxorubicin-induced cardiomyopathy (DIC). Currently, there are two methods of IP injection. Both methods have limitations when handling toxic compounds such as doxorubicin, which result in side effects manifesting as severe damage to the body shape and fish death. While these shortcomings could be overcome by extensive investigator training, a new IP injection method that has minimal side effects is desirable. Here, a unique IP injection method that is able to handle toxic compounds is reported. Consistently reduced cardiac function can result without incurring significant fish death. The technique can be easily mastered by researchers who have minimal experience with adult zebrafish.
Introduction
Zebrafish (Danio rerio) has gained attention as an experimental model for studying human diseases because this animal encompasses high gene and organ homology to humans, external fertilization, ease of genetic manipulation, and body transparency into early maturity, which facilitates a myriad of imaging applications1. Unlike the straightforward process of delivering drugs directly to the water for zebrafish embryos and larvae, administering drugs to adult zebrafish presents a more intricate and challenging endeavor2.
In adult fish, compounds can be delivered through passive drug delivery techniques, such as direct administration into the water, or through oral drug delivery methods like gavaging2. Other approaches include coating fish food with the compounds and subsequently feeding the fish3, and direct administration of water-insoluble medications at a predetermined concentration, including retro-orbital or intraperitoneal injections4,5. Intraperitoneal administration is preferred for in vivo studies of disease models due to its distinct pharmacokinetic advantages6. This method provides a high drug concentration and an extended half-life within the peritoneal cavity, offering an effective route for drug delivery7,8. The approach is commonly utilized in research settings to ensure optimal drug absorption and distribution 9. While injection-based methods prove efficient for single delivery, prolonged and repeated injections often lead to body damage and chronic infection2.
Currently, there are two methods of IP injection in adult zerbafish4,10. However, both methods have limitations when delivering toxic compounds like doxorubicin, leading to severe damage to the body shape and fish mortality. The side effects can significantly complicate data interpretation. Although these challenges may be addressed with extensive training10, there is a clear need for a new IP injection method that minimizes side effects.
Here, our goal is to develop a new method of IP injection optimized for the effective delivery of doxorubicin into adult zebrafish, facilitating the generation of reliable doxorubicin-induced cardiomyopathy (DIC) models with minimized body damage and associated mortality.
Protocol
All procedures conducted were approved by the Mayo Clinic Institutional Animal Care and Use Committee, adhering to the standards outlined in the 'Guide for the Care and Use of Laboratory Animals' (National Academies Press, 2011). All zebrafish in the study belong to the Wild Indian Karyotype (WIK) strain. The details of the reagents and the equipment used for the study are listed in the Table of Materials.
1. Preparation and storage of doxorubicin stock solution
- Obtain the doxorubicin stock from a commercial source.
NOTE: Doxorubicin is light-sensitive, so acquire it in powder form and store it in opaque containers to protect it from exposure to light. Perform all steps for the Dox powder preparation within a chemical hood. - Completely dissolve the Dox powder in distilled water and prepare a stock solution with a final concentration of 5 mg/mL.
- Aliquot the stock by dividing it into 1.5 mL tubes.
- Wrap the tubes in aluminum foil to shield them from exposure to light.
- Store the aliquot Dox solution at 4 °C for short-term storage (<1 month), or -20 °C for long-term storage10.
2. Grouping the fish according to their body weight
- Group the fish with a BW difference of less than 10% together for subsequent injection.
NOTE: To save effort at this phase, fish with less than a 10% BW variance are categorized as the same size. - Allow the fish to fast for 24 h prior to injection.
- Anesthetize the fish using embryo water containing 0.16 mg/mL tricaine for 1 min.
- Take the fish out of the water with tricaine and dab both sides of the fish body with clean filter paper to remove excess water.
- Measure and record the BW of each fish, then promptly return the fish to a recovery tank filled with fresh system water.
NOTE: Dox injection was performed on fish after reaching 3 months of age. In this study, researchers utilized 3-month to 10-month-old fishes. The BWs of mature WIK strain zebrafish may vary from 0.2 g to 0.5 g. Prolonged anesthesia lasting for more than 5 min, followed by a Dox injection, led to high fish mortality.
3. Preparation of the needle and station for injection
- Determine the injection volume of Dox stock solution (e.g., 5 mg/mL) needed for each fish based on the average body weight to achieve the targeted dose of 20 µg/g.
- Use the following formula to calculate the injection volume:
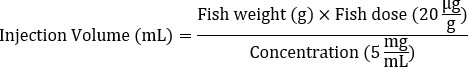
- Add 1x Hank's Balanced Salt Solution (HBSS) to dilute the Dox solution calculated in step 1 for injection, reaching a total volume of 5 µL.
NOTE: Utilize bulk solution for each group of fish based on their BW and include an additional 3 fish in each group to ensure that there is no shortage of solution for injection during the course of the experiment. - Gently tap the tube and then briefly microcentrifuge at top speed to collect the solution at room temperature for 10 s.
- Place the prepared solution on ice and shield it from exposure to light.
- Place a clean 100 mm Petri dish with a sponge underneath a dissecting microscope, then adjust the focus.
NOTE: The sponge contains a 4 cm long groove. The elastic retraction of the sponge will provide the fish with support and keep the fish body in position. The sponge can be reused. - Equip a 10 µL micro-syringe with a 34 G beveled needle.
- Flush the needle with 1x HBSS buffer to eliminate any bubbles and clear potential blockages from the syringe.
- Measure 5 µL of the solution prepared in step 4 for the injection.
4. IP Dox injection procedure
- Put the adult fish in water with 0.16 mg/mL tricaine for 1 min to induce a state of unconsciousness.
- Position the fish in the groove of the embedded sponge with the abdomen facing upward (Figure 1A).
- Insert the needle with an angle near 0°, starting from the midpoint of the pectoral fin towards the posterior side of the cardiac cavity (Figure 1B).
NOTE: Avoid any contact with the heart during the procedure. - Direct the needle towards the tail and go beneath the silver skin.
NOTE: Position the needle in close proximity to the silver skin, taking care to avoid any scratching or piercing. - Monitor the needle tip within the abdominal cavity throughout the entire operation (Figure 1C).
NOTE: Avoid damaging the liver, intestines, swimming bladder, and other organs. Ensure that the needle reaches the end of the intestine, near the cloacal foramen. - Gradually and evenly dispense the 5 µL Dox solution, then slowly withdraw the needle along the original path to prevent any leakage (Figure 1D).
- Monitor the abdominal cavity for the presence of Dox by observing a red coloration of Dox solution (Figure 1E).
- Swiftly move the injected fish to a clean crossing tank filled with fresh system water to help the fish recuperate.
NOTE: Between injections, flush the needle once with 1x HBSS buffer.
5. Post-injection fish management
- Return the fish back to the system with circulation after the injection.
- Fast all the injected fish for an additional 24 h to facilitate their recovery.
- During the first week, keep a close eye on the fish. Remove the deceased fish as soon as possible to avoid infecting the other fish.
NOTE: Fish fatalities within the initial 24 h are likely due to physical injuries from the injection or prolonged anesthesia. Record fish numbers to generate a survival curve. - Perform echocardiography to phenotype the Dox-injected fish at 56 days post-injection11.
NOTE: Ensure uniformity in conditions and procedures for the corresponding control group injected with HBSS solution.
Results
Previously, two intraperitoneal (IP) methods have been employed for the administration of doxorubicin in adult zebrafish4,10. In method I, also known as the Classic IP injection method as described by Kinkel et al.4, the needle was inserted at a 45° angle to the midline between the pelvic fins with the abdomen facing upward. In method II, or the Alternative IP injection method as described by Ma et al.10, the n...
Discussion
Different from the two existing IP injection methods4,10, the new IP injection method is characterized by the following distinct features.Firstly, a unique needle penetration angle is used (close to zero); secondly, the needle penetrates the fish via a unique location, i.e., a natural hole on the ventral surface of a fish, which would facilitate the injection; and finally, the movement of the needle is from anterior to posterior. These adjustments effect...
Disclosures
None.
Acknowledgements
This study was supported by the NIH (HL107304 and HL081753) and the Mayo Foundation (Center for Biomedical Discovery and Cardiovascular Research Center) to X.X. J.L. is funded by the Fundamental Research Funds for the Central Universities of Central South University, No. 56021702. Special thanks to Beninio Gore and Quentin Stevens for managing the zebrafish facility.
Materials
| Name | Company | Catalog Number | Comments |
| 10 μL NanoFil-syringe | World Precision Instruments, Inc | NANOFIL | injection tool |
| 34 G needle | World Precision Instruments, Inc | NI34BV-2 | injcetion tool |
| 60 mm Petri dish | fisher scientific/fisherbrand | FB0875713A | placing the sponge |
| Dissecting microscope | Nikon | SMZ800 | Injceting the Dox |
| Doxorubicin hydrochloride | Sigma | D1515-10MG | drug for creating DIC model |
| Echocardiography | VISUAL SONICS | Vevo 3100 | measuring cardiac function |
| Foam Sponge | Jaece Industries | L800-D | placing the fish |
| Hank's balanced salt solution (HBBS) | Thermo Fisher | 14025076 | Vehicle for Dox |
| Microcentrifuge | southernlabware | MyFuge/C1012 | collect the Dox solution |
| Precision Balance Scale | Torbal | AD60 | Digital scales |
| Tricaine | Argent | MS-222 | Anesthetizing fish |
| Tube | Eppendorf | 1.5 mL | storage |
| vevo LAB software | FUJIFILM VISUAL SONICS | 5.6.0 | quantification of the heart |
References
- Tavares, B., Lopes, S. S. The importance of Zebrafish in biomedical research. Acta Medica Portuguesa. 26 (5), 583-592 (2013).
- Dang, M., Henderson, R. E., Garraway, L. A., Zon, L. I. Long-term drug administration in the adult zebrafish using oral gavage for cancer preclinical studies. Disease Model Mech. 9 (7), 811-820 (2016).
- Sciarra, J. B., Tyler, A., Kolb, A. A gelatin-based diet for oral dosing juvenile to adult zebrafish (Danio rerio). Lab Animal Sci Prof. , 32-35 (2014).
- Kinkel, M. D., Eames, S. C., Philipson, L. H., Prince, V. E. Intraperitoneal injection into adult zebrafish. J Vis Exp. (42), e2126 (2010).
- Pugach, E. K., Li, P., White, R., Zon, L. Retro-orbital injection in adult zebrafish. J Vis Exp. (34), e1645 (2009).
- Liu, J., et al. Intraperitoneally delivered mesenchymal stem cells alleviate experimental colitis through THBS1-mediated induction of IL-10-competent regulatory B cells. Front Immunol. 13, 853894 (2022).
- De Smet, L., Ceelen, W., Remon, J. P., Vervaet, C. Optimization of drug delivery systems for intraperitoneal therapy to extend the residence time of the chemotherapeutic agent. The Scientific World Journal. 2013, 720858 (2013).
- Dakwar, G. R., et al. Nanomedicine-based intraperitoneal therapy for the treatment of peritoneal carcinomatosis-Mission possible. Adv Drug Deliv Rev. 108, 13-24 (2017).
- Al Shoyaib, A., Archie, S. R., Karamyan, V. T. Intraperitoneal route of drug administration: should it be used in experimental animal studies. Pharmaceutical Res. 37, 1-17 (2020).
- Ma, X., Ding, Y., Wang, Y., Xu, X. A doxorubicin-induced cardiomyopathy model in adult zebrafish. J Vis Exp. (136), e57567 (2018).
- Wang, L. W., et al. Standardized echocardiographic assessment of cardiac function in normal adult zebrafish and heart disease models. Disease Model Mech. 10 (1), 63-76 (2017).
- Christidi, E., Brunham, L. R. Regulated cell death pathways in doxorubicin-induced cardiotoxicity. Cell Death Dis. 12 (4), 339 (2021).
- Zhu, W., Shou, W., Payne, R. M., Caldwell, R., Field, L. J. A mouse model for juvenile doxorubicin-induced cardiac dysfunction. Pediatric Res. 64 (5), 488-494 (2008).
- Podyacheva, E. Y., Kushnareva, E. A., Karpov, A. A., Toropova, Y. G. Analysis of models of doxorubicin-induced cardiomyopathy in rats and mice. A modern view from the perspective of the pathophysiologist and the clinician. Frontiers Pharmacol. 12, 670479 (2021).
- Chaoul, V., et al. Assessing drug administration techniques in zebrafish models of neurological disease. Int J Mol Sci. 24 (19), 14898 (2023).
- Lu, X., Lu, L., Gao, L., Wang, Y., Wang, W. Calycosin attenuates doxorubicin-induced cardiotoxicity via autophagy regulation in zebrafish models. Biomed Pharmacother. 137, 111375 (2021).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved