A subscription to JoVE is required to view this content. Sign in or start your free trial.
Fabrication of Antibacterial Graphene Oxide/Copper Nanocomposites
In This Article
Summary
Herein, we introduce graphene oxide/copper (GO/Cu) nanocomposites as an antibacterial nanomaterial. The antibacterial effectiveness of the GO/Cu nanocomposites was evaluated against both antibiotic-resistant gram-positive and gram-negative bacteria.
Abstract
Antibiotics are currently the most used antibacterial treatment for killing bacteria. However, bacteria develop resistance when continually overexposed to antibiotics. Developing antimicrobial agents that can replace existing antibiotics is essential because antibiotic-resistant bacteria have resistance mechanisms for all current antibiotics and can promote nosocomial infections. To address this challenge, in this study, we propose graphene oxide/copper (GO/Cu) nanocomposites as antibacterial materials that can replace the existing antibiotics. GO/Cu nanocomposites are characterized by transmission electron microscopy and scanning electron microscopy. They show that copper (Cu) nanoparticles are well-grown on the graphene oxide sheets. Additionally, a microdilution broth method is used to confirm the efficacy of the antimicrobial substance against methicillin-resistant Staphylococcus aureus (MRSA) and Pseudomonas aeruginosa (P. aeruginosa), which are frequently implicated in nosocomial infections. Specifically, 99.8% of MRSA and 84.7% of P. aeruginosa are eliminated by 500 µg/mL of GO/Cu nanocomposites. Metal nanocomposites can eradicate antibiotic-resistant bacteria by releasing ions, forming reactive oxygen species, and physically damaging the bacteria. This study demonstrates the potential of antibacterial GO/Cu nanocomposites in eradicating antibiotic-resistant bacteria.
Introduction
Bacterial infections have a significant impact on public health. Pathogenic bacteria, in particular, can evade the body's protective mechanisms and cause diseases1. Antibiotics are widely used to treat bacterial infections. However, the inappropriate use of antibiotics has precipitated the emergence of antibiotic-resistant bacteria. Currently, nosocomial infections attributable to antibiotic-resistant bacteria have caused notable complications in healthcare facilities2. Unfortunately, bacteria have resistance mechanisms for all current antibiotics3. Therefore, developing new antibiotics is essential, though there is also a high probability that resistance mechanisms will emerge.
Metal nanoparticles have emerged as promising agents for combating antibiotic-resistant bacteria because of their effective antibacterial properties4,5,6. It is difficult for bacteria to develop resistance mechanisms against metal nanoparticles because they do not bind to any specific bacterial receptors7. In particular, silver, copper, and zinc oxide nanoparticles are the focus of extensive research because of their remarkable antibacterial efficacy8,9,10,11,12,13. The toxicity of metal nanoparticles toward bacteria is attributed to three primary mechanisms: 1) the release of metal ions, 2) the generation of reactive oxygen species (ROS), such as •OH and •O2−, and 3) physical interaction and attachment4,14.
In this study, graphene oxide/copper (GO/Cu) nanocomposites were developed as an antimicrobial agent. Copper (Cu) efficiently kills pathogens that encounter its surface and interferes with bacterial gene replication. However, it is an essential microelement in the human body, and it is less toxic to mammalian cells because it has homeostatic mechanisms that regulate the concentrations of Cu inside the cells4. When Cu nanoparticles are oxidized, they generate Cu ions, which have a relatively high affinity for negatively charged bacteria15,16. Cu ions bind to the cellular structures (e.g., proteins, membranes, and DNA), disrupting cell functions17. By synthesizing Cu nanoparticles on the surface of graphene oxide (GO), the release rate of metal ions can be controlled18,19,20. GO also shows an antibacterial effect by preventing bacterial adhesion with a rough surface through oxidative stress or by forming ROS21,22. Moreover, GO/Cu nanocomposites release more ROS than GO through the chemical action of Cu2+, which damages bacterial proteins and DNA, leading to the death of bacteria23,24.
This paper outlines the protocol for synthesizing GO/Cu nanocomposites and introduces an antimicrobial testing approach against clinical bacteria MRSA and P. aeruginosa, the most common multidrug-resistant bacteria strains that cause nosocomial infections25. This protocol aims to introduce a facile chemical reduction method for synthesizing GO/Cu nanocomposites that can impede the growth of antibiotic-resistant bacteria. We use copper(II) chloride (CuCl2) and sodium borohydride (NaBH4) as a precursor and reducing agent, respectively. The protocol also describes the specifics of applying these nanocomposites to bacteria using the microdilution broth method. To exclude errors due to the absorption interference of metal nanoparticles and make accurate evaluations, the antibacterial ability is evaluated using the colony counting method.
Protocol
1. Preparation of the GO/Cu nanocomposites
NOTE: The size and morphology of Cu nanoparticles that grow on the GO nanosheets are determined by the degree of GO oxidation, the concentration of the Cu precursor, and the concentration of the reducing agent26.
- Prepare 10 mL of 1 mg/mL GO suspension in a glass bottle. Sonicate the GO suspension for 1 h until GO is well dispersed in distilled (DI) water.
- Prepare a 20 mM CuCl2 solution in a glass vial. Sonicate the CuCl2 solution until CuCl2 is well dispersed in DI water.
- Add 10 mL of the 20 mM CuCl2 solution to the GO solution and sonicate the mixture at 70 °C for 1 h.
- Prepare a 20 mM NaBH4 solution.
CAUTION: This procedure must be conducted in a chemical fume hood. A redox reaction occurs as soon as the reducing agent NaBH4 is introduced into water. Prepare the NaBH4 solution immediately before the experiment. - Preparing the mixture of NaBH4, GO, and CuCl2
- Add 20 mL of the 20 mM NaBH4 solution to the GO and CuCl2 mixture while stirring with a magnetic bar at 200 rpm. After adding the NaBH4 solution, stir continuously for 30 min.
CAUTION: This procedure must be conducted in a chemical fume hood.
- Add 20 mL of the 20 mM NaBH4 solution to the GO and CuCl2 mixture while stirring with a magnetic bar at 200 rpm. After adding the NaBH4 solution, stir continuously for 30 min.
- Centrifugation of the mixture
- Transfer the mixture from step 1.5.1 to a centrifuge tube. Centrifuge the solution at 23,000 × g for 10 min at room temperature. Discard the supernatant.
CAUTION: When operating the centrifuge, always maintain balance by placing tubes of equal weight.
- Transfer the mixture from step 1.5.1 to a centrifuge tube. Centrifuge the solution at 23,000 × g for 10 min at room temperature. Discard the supernatant.
- Resuspend the GO/Cu nanocomposite mixture with 10 mL of DI water and sonicate them to enable the precipitate to disperse evenly throughout the solution.
- Centrifuge the solution at 23,000 × g for 10 min at room temperature; then remove the supernatant.
- Repeat steps 1.7-1.8 once more to remove the unreacted chemicals.
- Add 1 mL of distilled water to the GO/Cu nanocomposites and sonicate the mixture to disperse the sediment evenly throughout the liquid phase.
NOTE: Collect all GO/Cu nanocomposite solutions in one conical tube. - Freeze-dry the solution at −60 °C under a vacuum overnight until the GO/Cu nanocomposites are completely dried and obtain GO/Cu nanocomposite powder.
- Store the GO/Cu nanocomposite powder at −20 °C until use.
2. Preparation of bacteria for the antibacterial test
CAUTION: This procedure must be conducted in a biological safety cabinet with an alcohol lamp. Bacterial waste must be autoclaved before disposal. When handling antibiotic-resistant bacteria, gloves, gowns, and masks must be worn, and hands must be washed using soap or hand sanitizer after the experiment. Always thoroughly disinfect; if contamination occurs in the experimental area, immediately disinfect it with 70% ethanol.
- Bacterial culture media preparation
- Mix 20 g of tryptic soy agar and 500 mL of distilled water in a flask. Sterilize the mixture in an autoclave at 121 °C for 15 min.
- Prepare the agar plate by pouring 15 mL of the agar solution into a Petri dish before the agar hardens. Store the agar plate at 4 °C until use.
NOTE: This must be done quickly before the agar solution hardens. To prevent contamination, sterilize the agar plate with UV for 15 min during hardening. - Mix 15 g of tryptic soy broth (TSB) and 500 mL of distilled water in a flask. Sterilize the mixture in an autoclave at 121 °C for 15 min.
- Aliquot 40-50 mL of the broth solution into a 50 mL conical tube. Store the broth at 4 °C until use.
NOTE: To prevent contamination, sterilize the aliquoted broth with UV for 15 min.
- Bacterial culture
- Inoculate the MSRA or P. aeruginosa stock solution on the agar plate using a loop. Incubate the agar plate at 37 °C using a shaking incubator for 24 h.
- Inoculate one colony of the bacteria into 10 mL of the broth using a loop. Incubate the broth at 37 °C using a shaking incubator at 200 rpm for 24 h.
- Repeat steps 2.2.1-2.2.2 once more to obtain bacteria with the same characteristics.
- After 24 h, serially dilute the bacterial solution tenfold using sterile distilled water. Inoculate 100 µL of the diluted bacterial suspension onto the agar plate and spread it with a spreader. Incubate the agar plate at 37 °C using a shaking incubator for 24 h.
NOTE: The dilution level is dependent on the bacteria. Aim to prepare less than 100 colonies in an agar plate. - Count the bacterial colonies after the 24 h incubation to determine the colony-forming unit (CFU) using equation (1).
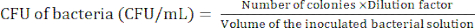 (1)
(1) - Repeat steps 2.2.4-2.2.5 at least 3x to confirm whether the CFU of the bacteria is relatively constant.
3. Antibacterial test using the microdilution broth method
- Day 1) Inoculate one colony of the bacteria from the agar plate into 10 mL of the broth using a loop. Incubate the broth at 37 °C using a shaking incubator at 200 rpm for 24 h.
- Day 2) Prepare the GO/Cu nanocomposite mixture in at least three concentrations using Dulbecco's phosphate-buffered saline (DPBS). Sonicate the GO/Cu nanocomposite suspension until the GO/Cu nanocomposites are well dispersed in DPBS.
NOTE: In this protocol, 500, 250, 125, and 62.5 µg/mL of the GO/Cu nanocomposite mixture were tested. - Prepare the control solutions. The negative control is DPBS, and the positive control is 1% of a penicillin/streptomycin solution in DPBS that will kill the bacteria.
- Add 100 µL of the GO/Cu nanocomposite suspension and the control solutions into 96-well plates. Add all samples in triplicate.
NOTE: Sterilize samples with UV for 15 min before applying them to bacteria. - Based on the CFU after 24 h of incubation, dilute bacterial suspension using TSB to 1 × 106 CFU/mL.
NOTE: The initial MRSA and P. aeruginosa culture suspension concentrations in our study, according to step 2.2.5, are 4.5 × 109 CFU/mL and 3 × 109 CFU/mL, respectively. Bacterial concentrations of 1 × 106 CFU/mL are obtained by diluting the cultures 4,500x and 3,000x, respectively. - Inoculate 100 µL of the 1 × 106 CFU/mL diluted bacterial suspension into the sample wells in the 96-well plates. Incubate the 96-well plates at 37 °C using a shaking incubator at 200 rpm for 24 h.
NOTE: The final concentration of bacteria is 5 × 105 CFU/mL after mixing with 100 µL of sample. - Day 3) Mix the sample and bacterial suspensions vigorously with a 200 µL micropipette tip. Serially dilute the sample-bacteria mixture tenfold with sterile distilled water.
NOTE: The dilution level is dependent on the bacteria. Aim to prepare less than 100 colonies in an agar plate. - Inoculate 100 µL of the diluted bacterial suspension onto the agar plate and spread it with a spreader. Incubate the agar plate at 37 °C using a shaking incubator for 24 h.
- Day 4) Count the bacterial colonies and determine the CFU values to confirm the antibacterial activity of the GO/Cu nanocomposites using equation (2).
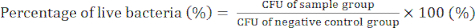 (2)
(2)
Results
Transmission electron microscopy (TEM), scanning electron microscopy (SEM), and energy-dispersive X-ray spectroscopy (EDS) analyses were performed to confirm the formation of GO/Cu nanocomposites. Figure 1A,B show that heterogeneous Cu nanoparticles were grown on the GO sheets. As shown in Figure 1C, the EDS mapping confirmed that the particles on the GO sheet were Cu nanoparticles.
Discussion
Herein, we report a low-cost and simple method for preparing GO nanosheets deposited with Cu nanoparticles, which would be a potentially efficient method for eradicating antibiotic-resistant bacteria. The critical step in synthesizing GO/Cu nanocomposites is thoroughly dispersing GO and CuCl2 in the solution and maintaining an elevated temperature while mixing them. Further, the redox step must be conducted quickly because the reducing agent causes a reduction reaction as soon as it comes in contact with water...
Disclosures
Dr. Jonghoon Choi is the CEO/Founder, and Dr. Yonghyun Choi is the CTO of the Feynman Institute of Technology at the Nanomedicine Corporation.
Acknowledgements
This research was supported by the Chung-Ang University research grant in 2022 (during Professor Jonghoon Choi's sabbatical leave). This research was also supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education (No. RS-2023-00275006), and the Korea Environment Industry & Technology Institute (KEITI) grant funded by the Korea Ministry of Environment (MOE) (No. 2022002980003). Clinical MRSA and P. aeruginosa were obtained from Chung-Ang University Hospital.
Materials
| Name | Company | Catalog Number | Comments |
| Antibiotic-Antimycotic | Gibco | 15240062 | |
| Clinical MDR bacterial strains | Chung-Ang University Hospital (Seoul, South Korea) | ||
| Copper(II) chloride dihydrate | Duksan | 10125-13-0 | |
| Field Emission Scanning Electron Microscope (FE-SEM) | Carl Zeiss | SIGMA | |
| Graphene oxide | Sigma | 796034 | |
| Sodium Borohydride | Sigma | 71320 | |
| Transmission Electron Microscopy (TEM) | JEOL | JEM-2100 | |
| Tryptic Soy Agar | BD difco | 236950 | |
| Tryptic Soy Broth | BD difco | 211825 |
References
- Doron, S., Gorbach, S. L. Bacterial infections: Overview. International Encyclopedia of Public Health. , 273-282 (2008).
- Van Duin, D., Paterson, D. L. Multidrug-resistant bacteria in the community: An update. Infect Dis Clin North Am. 34 (4), 709-722 (2020).
- Blair, J. M. A., Webber, M. A., Baylay, A. J., Ogbolu, D. O., Piddock, L. J. V. Molecular mechanisms of antibiotic resistance. Nat Rev Microbiol. 13 (1), 42-51 (2015).
- Sánchez-López, E., et al. Metal-based nanoparticles as antimicrobial agents: An overview. Nanomaterials (Basel). 10 (2), 292 (2020).
- Makabenta, J. M. V., et al. Nanomaterial-based therapeutics for antibiotic-resistant bacterial infections. Nat Rev Microbiol. 19 (1), 23-36 (2021).
- Liang, X., Govindaraju, S., Yun, K. Dual applicability of polyaniline coated gold nanorods: A study of antibacterial and redox activity. BioChip J. 12 (2), 137-145 (2018).
- Niño-Martínez, N., Salas Orozco, M. F., Martínez-Castañón, G. A., Torres Méndez, F., Ruiz, F. Molecular mechanisms of bacterial resistance to metal and metal oxide nanoparticles. Int J Mol Sci. 20 (11), 2808 (2019).
- Kim, J., Choi, Y., Park, J., Choi, J. Gelatin-gallic acid microcomplexes release go/cu nanomaterials to eradicate antibiotic-resistant microbes and their biofilm. ACS Infect Dis. 9 (2), 296-307 (2023).
- Jang, J., et al. Development of antibiofilm nanocomposites: Ag/cu bimetallic nanoparticles synthesized on the surface of graphene oxide nanosheets. ACS Appl Mater Interfaces. 12 (32), 35826-35834 (2020).
- Jang, J., Choi, Y., Tanaka, M., Choi, J. Development of silver/graphene oxide nanocomposites for antibacterial and antibiofilm applications. J Ind Eng Chem. 83, 46-52 (2020).
- Bankier, C., et al. Synergistic antibacterial effects of metallic nanoparticle combinations. Sci Rep. 9 (1), 16074 (2019).
- Mendes, C. R., et al. Antibacterial action and target mechanisms of zinc oxide nanoparticles against bacterial pathogens. Sci Rep. 12 (1), 2658 (2022).
- Yoon, S., et al. Biologically benign multi-functional mesoporous silica encapsulated gold/silver nanorods for anti-bacterial applications by on-demand release of silver ions. BioChip J. 13 (4), 362-369 (2019).
- Slavin, Y. N., Asnis, J., Häfeli, U. O., Bach, H. Metal nanoparticles: Understanding the mechanisms behind antibacterial activity. J Nanobiotechnology. 15 (1), 65 (2017).
- Ismail, N. A., et al. Sonochemical synthesis of a copper reduced graphene oxide nanocomposite using honey and evaluation of its antibacterial and cytotoxic activities. Front Mol Biosci. 9, 995853 (2022).
- Tu, Y., et al. Remarkable antibacterial activity of reduced graphene oxide functionalized by copper ions. Adv Funct Mater. 31 (13), 2008018 (2021).
- Li, M., et al. Toward a molecular understanding of the antibacterial mechanism of copper-bearing titanium alloys against staphylococcus aureus. Adv Healthc Mater. 5 (5), 557-566 (2016).
- Yang, Z., et al. Long-term antibacterial stable reduced graphene oxide nanocomposites loaded with cuprous oxide nanoparticles. J Colloid Interface Sci. 533, 13-23 (2019).
- Yang, Y., et al. Graphene oxide/copper nanoderivatives-modified chitosan/hyaluronic acid dressings for facilitating wound healing in infected full-thickness skin defects. Int J Nanomedicine. 15, 8231-8247 (2020).
- Mao, M., et al. Graphene oxide-copper nanocomposites suppress cariogenic streptococcus mutans biofilm formation. Int J Nanomedicine. 16, 7727-7739 (2021).
- Ravikumar, V., Mijakovic, I., Pandit, S. Antimicrobial activity of graphene oxide contributes to alteration of key stress-related and membrane bound proteins. Int J Nanomedicine. 17, 6707-6721 (2022).
- Afreen, G., Shoeb, M., Upadhyayula, S. Effectiveness of reactive oxygen species generated from rgo/cds qd heterostructure for photodegradation and disinfection of pollutants in waste water. Mater Sci Eng C. 108, 110372 (2020).
- Yang, F., et al. Fabrication of graphene oxide/copper synergistic antibacterial coating for medical titanium substrate. J Colloid Interface Sci. 638, 1-13 (2023).
- Rajapaksha, P., et al. Antibacterial properties of graphene oxide-copper oxide nanoparticle nanocomposites. ACS Appl Bio Mater. 2 (12), 5687-5696 (2019).
- Sachdeva, C., Satyamoorthy, K., Murali, T. S. Microbial interplay in skin and chronic wounds. Curr Clin Microbiol Rep. 9 (3), 21-31 (2022).
- Ramos, M. K., Zarbin, A. J. G. Graphene/copper oxide nanoparticles thin films as precursor for graphene/copper hexacyanoferrate nanocomposites. Appl Surf Sci. 515, 146000 (2020).
- Fivenson, E. M., et al. A role for the gram-negative outer membrane in bacterial shape determination. PNAS. 120 (35), e2301987120 (2023).
- Wu, C., et al. Copper-containing mesoporous bioactive glass scaffolds with multifunctional properties of angiogenesis capacity, osteostimulation and antibacterial activity. Biomater. 34 (2), 422-433 (2013).
- Zhang, W., et al. Graphene oxide-copper nanocomposite-coated porous cap scaffold for vascularized bone regeneration via activation of hif-1α. Adv Healthc Mater. 5 (11), 1299-1309 (2016).
- Labauve, A. E., Wargo, M. J. Growth and laboratory maintenance of pseudomonas aeruginosa. Curr Protoc Microbiol. 25 (1), 6E.1.1-6E.1.8 (2012).
- Lin, J., et al. The icmf3 locus is involved in multiple adaptation- and virulence-related characteristics in pseudomonas aeruginosa pao1. Front Cell Infect Microbiol. 5, 70 (2015).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionExplore More Articles
This article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved