A subscription to JoVE is required to view this content. Sign in or start your free trial.
A Visual Approach for Inducing Dolichoectasia in Mice to Model Large Vessel-Mediated Cerebrovascular Dysfunction
In This Article
Summary
We demonstrate chemically inducing large blood vessel dilatation in mice as a model for investigating cerebrovascular dysfunction, which can be used for vascular dementia and Alzheimer's disease modeling. We also demonstrate visualizing the vasculature by injecting silicone rubber compound and providing clear visual guidance for measuring changes in blood vessel size.
Abstract
The blood-brain (BBB) is a crucial system that regulates selective brain circulation with the periphery, as an example, allowing necessary nutrients to enter and expel excessive amino acids or toxins from the brain. To model how the BBB can be compromised in diseases like vascular dementia (VaD) or Alzheimer's disease (AD), researchers developed novel methods to model vessel dilatation. A compromised BBB in these disease states can be detrimental and result in the dysregulation of the BBB leading to untoward and pathological consequences impacting brain function. We were able to modify an existing technique that enabled us to inject directly into the Cisterna magna (CM) to induce dilatation of blood vessels using elastase, and disrupt the tight junctions (TJ) of the BBB. With this method, we were able to see various metrics of success over previous techniques, including consistent blood vessel dilatation, reduced mortality or improved recovery, and improving the fill/opacifying agent, a silicone rubber compound, delivery for labeling blood vessels for dilatation analysis. This modified minimally invasive method has had promising results, with a 19%-32% increase in sustained dilatation of large blood vessels in mice from 2 weeks to 3 months post-injection. This improvement contrasts with previous studies, which showed increased dilatation only at the 2 week mark. Additional data suggests sustained expansion even after 9.5 months. This increase was confirmed by comparing the diameter of blood vessels of the elastase and the vehicle-injected group. Overall, this technique is valuable for studying pathological disorders that affect the central nervous system (CNS) using animal models.
Introduction
Microvascular endothelial cells that line the cerebral capillaries are the main components for forming the blood-brain barrier (BBB)1 which plays a crucial role in regulating what enters or leaves the brain circulation with the periphery. Essential nutrients needed for nervous tissue are permitted to enter the BBB, while some essential amino acids like glutamate are expelled from the brain, as high concentrations can cause permanent neuroexcitatory damage to brain tissue2. Under normal physiological conditions, the BBB limits the amount of plasma proteins like albumin3,4 and prothrombin from entering the brain since those can have detrimental effects5,6,7. Finally, the BBB protects the brain from neurotoxins that are circulating in the periphery, such as xenobiotics from food or the environment1. Overall, damage to brain tissues is irreversible, and aging that correlates with low levels of neurogenesis8 highlights the importance of the BBB in protecting and preventing any factors from accelerating the neurodegenerative process.
In dolichoectasia (or large blood vessel dilatation), a decrease in vessel elasticity is observed, which results in vessels undergoing morphological changes, thus rendering them dysfunctional9 and leading to reduced blood flow in the brain. This reduction in blood flow subsequently diminishes oxygen and glucose supply, ultimately triggering damage to the BBB through the activation of reactive astrocytes10. When the internal elastin lamina of vessels is damaged from dolichoectasia11, repeated stimulation of the vascular endothelial growth factor (VEGF) is necessary for angiogenesis. This can lead to the formation of leaky vessels and ultimately result in pathological angiogenesis, characterized by the development of defective vessels12. During pathological angiogenesis, when blood vessels become defective, a compensatory mechanism appears to restore vessel integrity by upregulating tight junction proteins. However, this process can inadvertently disrupt the BBB when the structural integrity of a blood vessel is lost13. This may occur through further disrupting the BBB and promoting the production of amyloid plaque14. Additionally, leakage from the periphery can cause neuroinflammation15, which results in neuronal degeneration and subsequent memory loss.
Structurally the protection that the BBB provides is through the tight junctions that prevent xenobiotic agents from the blood entering the brain. When permitting certain substances to enter the brain, the BBB mainly does it through two major processes, passive diffusion, or specific channels (like ion channels and transporters)1. In AD, research has demonstrated that a dysfunctional vascular system plays a significant role in the progression of the condition12,13. The formation of amyloid-beta (Aβ) plaques and neurodegeneration can result from the breakdown of the BBB12,13 and disturbances in cerebral blood flow16. A reduction in cerebral blood flow can be seen in elderly individuals diagnosed with vascular dementia and AD17,18. Damage to the blood-brain barrier (BBB) along with a dysfunctional cerebral blood flow (CBF) can contribute to the increased production of Aβ concentration in the brain, accompanied by the infiltration of foreign materials from the peripheral circulation19.
To investigate the pathogenesis of neurological diseases like AD and vascular dementia (VaD), models are developed to replicate the disease. In vitro models are extensively used but lack the biological environment for extensive disease modeling like mixed cell population, thus necessitating the importance of in vivo models. Mice are commonly used due to their ease of genetic manipulation in generating human-like properties (e.g., pathology) in disease. With the progression that has been made so far, there is still a continued need for improved models to emulate disease phenotypes like large vessel dilatation and their role in AD. To this end, we saw an opportunity and modified a technique that involved the injection of elastase into the Cisterna magna of mice20,21. Elastase is an enzyme that has been shown to break down elastin in connective tissue22 and in surrounding tight junctions23. The Cisterna magna was chosen as the point of injection due to it being located directly above the circle of Willis, the largest blood vessel in the brain. By injecting elastase into the Cisterna magna, we can compromise the BBB and blood vessels by breaking down the tight junctions and inducing dilatation of blood vessels (circle of Willis)24,25. Combining this technique with the use of an AD mouse model of pathology, for improved understanding of the pathogenesis for the vascular component of AD, can provide valuable insights into the complex interactions and influences between these two distinct pathologies.
Previous studies have demonstrated instances where patients display both the pathological features of AD and VaD, a condition typically referred to as mixed dementia26,27. Thus, understanding the interconnected mechanisms between both conditions can offer a more comprehensive perspective on the progression and manifestation of these neurodegenerative disorders, enhances our comprehension of the underlying mechanisms and potential therapeutic strategies. To this end, we demonstrate the application of elastase in an AD pathology mouse model (AppNL-F) to identify vascular changes.
Protocol
AppNL-Fmice (3 months old) that express human amyloid plaque at a physiological level were used for this study though this system can be used with any rodent model. All animal procedures were approved by the Animal Care Committee of CAMH (Protocol #843) and were in accordance with the ethical standards of the Canadian Council on Animal Care guidelines. Mice were bred in-house and kept on a 12-h light-dark cycle with ad libitum access to chow and water.
1. Procedure for Cisterna magna (CM) injection
- Surgical procedure
- Place the mouse (APPNL-F, both sexes, 3 months) in an anesthesia induction chamber for 1 min.
NOTE: Flush the chamber with 5% isoflurane mixed with 1% oxygen for at least 1 min. - After the mouse is anesthetized, remove the animal from the chamber and place it on a fresh surgical drape and maintain anesthesia (isoflurane, 2.5%-3%) by placing a nasal cone over the nose.
NOTE: Check depth of anesthesia by pinching the toe to ensure no reflexes. - Apply ophthalmic ointment to prevent dryness. Then, subcutaneously inject 0.1 mL of bupivacaine (local anesthetic; 1-2 mg/kg 0.125%, with a dilution ratio of 1:2) to the incision site and Metacam (analgesic; 5 mg/kg, with a dilution ratio of 1:10). Keep the animal hydrated by injecting 0.5 mL of saline subcutaneously before doing the procedure to compensate for any potential blood loss during surgery.
- For a clear incision site, shave the neck at the base of the occipital bone, wipe down the surface with sterile PBS and transfer the subject to the stereotaxic frame in the prone position.
NOTE: Ensure that anesthesia is maintained through the stereotaxic nose cone. - Place the nose bar from the stereotaxic frame on top of the mouse to provide stability and ensure the upper teeth are fastened.
- Position the animal head at a 120° angle from the body to elevate and distend the nape to expose the surgical area of interest.
- Clean the surgical site with Betadine scrub 3x, 70% ethanol 3x, and betadine solution 1x with sterile 2 inch x 2 inch gauze.
NOTE: Gloves and instruments should be sterile before the procedure. A sterile drape should also be placed over the incision site. - Make a small incision (1 cm) using a scalpel.
NOTE: This should reveal the midline of the nape of the neck. - Carefully run the scalpel along the midline to cut through the muscles. Then, using forceps gently separate the muscles by pulling them apart to the left and right.
NOTE: This should reveal the Cisterna magna (inverted triangle) which is located beneath the base of the skull (Figure 1).
- Place the mouse (APPNL-F, both sexes, 3 months) in an anesthesia induction chamber for 1 min.
- Preparation of elastase
NOTE: The package of elastase (powder form) that was ordered contained 250 Units (U). The amount of the elastase to be injected in each animal is 2.5 µL. The amount of elastase that is supposed to be in the 2.5 µL should be 15 milliUnits mU.- First calculate the concentration of the solution to be injected.
Concentration = mass/ volume
= 15 mU/2.5 µL
= 6 mU/µL
Therefore, the concentration that is required is 6 mU/µL. - The quantity of the elastase is 250 U and the study works with milliUnits, so convert the units to milliUnits which gives 250000 mU.
- After having a mass of 250000 mU and concentration of 6 mU/uL, figure out the volume (PBS) that would require diluting the elastase and maintaining a concentration of 6 mU/uL.
- Using the same formula above, rearrange to solve for volume:
Volume = mass/Conc.
= 250000 mU/ 6 mU/µL
= 41666.67 µL - Since it is hard to pipette 41666.67 µL, simply convert it to milliliters by dividing it by 1000 to get 41.66667 mL to obtain a 1x solution.
- To make the solution more concentrated,increase it by 10x or 100x.
- Create a 10x solution since it is simpler to work with (see below).
10x solution ---------- 4.166667 mL (conc.)
- Place the 250 U in a 5 mL aliquot tube and add 4.167 mL of sterile PBS into the aliquot tube to make the 10x stock solution.
NOTE: Prepare the elastase inside a sterile biosafety cabinet to maintain sterility. - Pipette 10 µL of stock solution and place in 100 aliquot tubes giving 1000 µL of the 4.167 mL stock solution. Store the tubes at -20 °C.
- Store the remainder of stock solution in aliquot tubes at 100 µL per tube and store in freezer at -20 °C.
NOTE: The solutions are stable at -20 °C for up to 6 months with no significant loss of activity. In addition, it is important to note that excessive freezing and thawing of the elastase can reduce stability. Therefore, to prevent that from happening, the stock solution can be divided before freezing such that only one sample needs to be thawed at a time for injection. - After thawing one of the aliquot tubes that contains 10 µL of a 10x solution, add 90 µL of sterile PBS to get of a 100 µL 1x solution.
- If the aliquot tube that contains 100 µL of a 10x solution was thawed instead, simply add 900 µL of PBS to get 1000 µL, which is a 1x solution. Store either solution at 4 °C for 1 week.
- First calculate the concentration of the solution to be injected.
- Injecting into the Cisterna magna
NOTE: Clean the Hamilton syringe before loading with elastase by loading and aspirating with distilled water 5x, then ethanol 5x, and finally distilled water 5x.- Load the Hamilton syringe with 2.5 µL of elastase (concentration of 15 mU) and ensure there are no air bubbles.
- Slowly insert the syringe with the bevel facing upward into the center of the Cisterna magna.
NOTE: Avoid puncturing any blood vessels that run across the Cisterna magna. - After the puncture, remove the bevel. This will facilitate the release of a small amount of cerebrospinal fluid (CSF). Use a sterile cotton swab to absorb the excess CSF.
- Reinsert the bevel into the puncture site, slowly inject elastase or vehicle over 1 min into the Cisterna magna and leave the needle in place for 1 min to prevent any leakage of elastase.
- After removing the bevel, close the incision site with a non-absorbable surgical suture (#4-0 with 3/8" needle), and turn off the anesthetic.
- Remove the animal from the stereotaxic frame and place on a warm heat pad set at 37 °C until the subject recovers.
NOTE: Another dose of Metacam is recommended after surgery to help with pain during post-surgical recovery.
2. Silicone rubber compound injection and tissue harvest
- Anesthetize the animal with an overdose of avertin (1 mL, 125-250 mg/kg).
NOTE: Pinch the toe to ensure reflexes cease. - Place the animal on a diaper and secure all limbs with surgical tape with the chest cavity facing upward
- Open the chest cavity using forceps and scissors to expose the heart.
- Insert a 23 G blunt needle into the left ventricle of the heart and perfuse with PBS infused with heparin (100 U/mL) for 4 min to clear the blood vessels using a micro-perfusion pump.
- Replace the buffer bottle with one containing 4% paraformaldehyde (PFA)(made up in PBS) and perfuse for another 4 min.
- Prepare the silicone rubber compound (yellow) solution by combining 5 mL of compound with 5 mL of the diluent in a 50 mL tube. Mix thoroughly before use.
- Fill a 10 mL syringe with 10 mL of silicone rubber compo und. Attach the syringe to the hose connected to a 23 G blunt needle, and carefully inject the solution manually into the heart.
NOTE: Due to the high viscosity of the silicone rubber compound, it is important to use a larger barrel syringe or the pressure will be too great to push the plunger. - Remove the mouse's head after injection and store at 4 °C overnight allowing the silicone rubber compound to cure in the vessels.
- Next day, gently remove the skull of the mouse (preferably using fine forceps), extract the brain, and incubate in 4% PFA overnight at room temperature (RT).
- After a 24 h incubation in PFA, wash the brain in PBS 3x, place in 30% sucrose, and store at 4 °C.
3. Quantitative analysis
- To capture high-quality images of the Circle of Willis in the entire brain (Figure 3):
- Remove the brain from the sucrose solution. Thoroughly dry the brain using a paper towel to wick the surface moisture.
- Position the brain in a brain slicer. Place the brain slicer under a light microscope equipped with a camera.
- Focus the microscope to a clear, high-resolution resolution of the Circle of Willis.
- Use image analysis software with a 10 cm ruler reference to measure approximately 1 cm and note the pixel count. Set the 1 cm scale based on this measurement, then measure the basilar artery's thickness horizontally to find its diameter.
- Use a standard pixel densitometry software to analyze the image. Measure (in centimeters) five separate readings of the basilar artery using an image analysis software and calculate the average to get an accurate representation of the basilar artery diameter.
- Use a 2-way analysis of variance (ANOVA) to compare the average change in diameter between the elastase and control group.
- Calculate the percentage change in diameter using the formula below 21:
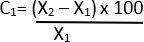
C = relative change
X1 = Initial value (Vehicle)
X2 = Final value (Elastase)
Consider P values less than 0.05 statistically significant. Perform all statistical analyses using a statistical analysis software.
- Calculate the percentage change in diameter using the formula below 21:
4. Removal of silicone rubber compound using alcohol dehydration
NOTE: It is important to dehydrate the brain to help remove the excess silicone rubber compound from the blood vessel which can potentially improve the quality of immunostaining.
- Wash the brains 3x with PBS to remove the sucrose.
- Place the brain in 25%, 50%, 75%, 95%, and absolute ethanol for 24 h at each concentration, for a total time of 5 days.
- After dehydration of tissue, place the tissue in methyl salicylate (conc. ≤100%) for 12-24 h. The ethanol and methyl salicylate will allow for the removal of excess silicone in the tissue.
- Wash the brain in PBS 3x to remove residual chemicals and leave overnight on a rocker at 4 °C in fresh PBS.
- Place the brain in sucrose and store at 4 °C prior to sectioning and immunostaining.
5. Immunohistochemical staining
- Sectioning of brains
- Prior to immunosectioning, allow the brain to settle at the bottom of the tube filled with sucrose.
- Freeze the brain on the microtome platform using optimal cutting temperature (OCT) compound surrounded by dry ice. Slice the brain into 40 µm sections using the microtome blade.
- Transfer each section into a 96-well plate containing cryoprotectant to prevent freezing using a fine tip paintbrush.
- Securely seal the 96-well plate with plastic wrap. Store the sealed plate in a -20 °C freezer.
- Immunofluorescence staining (using NeuN as an example)
- Place the sections in a 24-well plate. Perform three washes with PBS to eliminate the cryoprotectant.
- To prevent non-specific binding, incubate for 1 h at RT using a blocking solution containing 2% goat serum, 0.1% Triton-X 100, and 1% BSA.
- Prepare all components in PBS.
NOTE: The primary antibody used included monoclonal mouse anti-NeuN with a 1: 500 dilution. The secondary antibody used included polyclonal goat anti-mouse 568 with a 1:200 dilution. - Dilute the monoclonal mouse anti-NeuN in the blocking solution mentioned above.
- Add the diluted antibody to each well. Incubate overnight at 4 °C, shielded from light by foil.
- After the primary incubation, wash the sections 3x with PBS. Dilute the polyclonal goat anti-mouse 568 in the same blocking solution.
- Add the diluted antibody to each well. Incubate for 2 h at room temperature, shielded from light by foil.
- Wash the sections 3x with PBS after the secondary incubation.
- Prepare for mounting by slowly transferring sections onto slides using fine paintbrushes. Allow all sections to dry.
- Apply 125-150 µL of anti-fade mounting media once sections are dried. Gently place coverslips.
- Allow slides to dry for 24 h at RT in the dark, sealing edges with nail polish. Capture fluorescent images using imaging software.
Results
We were successful in locating the Cisterna magna beneath the occipital region of the skull after carefully positioning the mouse on the stereotaxic frame and dissecting the muscles. This anatomical structure, resembling an inverted triangle and highlighted in yellow, is situated beneath the base of the skull (Figure 1). To ensure precision and prevent any damage to the brain tissue, 1-2 mm of the Hamilton syringe bevel was gently inserted into the Cisterna magna.
Discussion
This article demonstrates an improved protocol for cerebrovascular dilatation, providing a precise and straightforward approach for elastase injection into the Cisterna magna of mice. This anatomical point serves as a direct gateway to the cerebrospinal fluid, offering a valuable avenue for the investigation of different neurological diseases. One of the main advantages of this modified technique is that injecting a single dose of elastase into the Cisterna magna of mice was able to cause and sustain dilatation of large ...
Disclosures
There is no conflict of interest.
Acknowledgements
This study was made possible by the invaluable contributions of Stephanie Tam who provided support in assisting with the surgeries. We extend our sincere gratitude for her help. The National Institutes of Health (AG066162) for support of this research.
Materials
| Name | Company | Catalog Number | Comments |
| 23 G catheter | University Medstore | 2546-CABD305145 | Needed for perfusion (https://www.uoftmedstore.com/index.sz) |
| Absolute ethanol | University Medstore | https://www.uoftmedstore.com/index.sz | For removing the microfil |
| Betadine scrub | # | https://www.pittsborofeed.com/products/betadine-surgical-scrub | Sterilization |
| Betadine solution | Amazon | https://www.amazon.ca/Povidone-Iodine-10-Topical-Solution-100ml/dp/B09DTKJGHW | Sterilization |
| Bupivacaine | Provided by animal facility | N/A | Analgesic |
| Clippers | BrainTree Scientific Inc | CLP-41590 | Shave fur |
| Cotton Q-tip | University Medstore | 1962 | For surgery (https://www.uoftmedstore.com/index.sz) |
| Elastase | Sigma-aldrich | E7885 | Used for the dilatation of blood vessel |
| Ethanol | University Medstore | 39752-P016-EAAN | Sterilization (https://www.uoftmedstore.com/index.sz) |
| Goat anti-mouse 568 | Invitrogen | A11004 | For staining mature neurons |
| Graphpad prism 10 | Graphpad prism 10 | https://www.graphpad.com/ | Statistical analysis software |
| Hamilton syringe | Sigma-aldrich | 28614-U | Injection elastase |
| Heat pad | Amazon | https://www.amazon.ca/iPower-Temperature-Controller-Terrarium-Amphibians/dp/B08L4DBFFZ | Maintain body temperature |
| ImageJ software | Fiji Imagej software | imagej.net (USA) | Image analysis software |
| Induction chamber | Provided by animal facility | N/A | Anesthesia induction |
| Metacam | Provided by animal facility | N/A | Analgesic |
| Methyl salicylate | Sigma-aldrich | M6752 | For removing the microfil |
| Microfil | Flow Tech, Carver, Massachusetts | https://www.flowtech-inc.com/order/ | Dye (yellow) |
| Mouse monoclonal anti-NeuN | Millipore Sigma | MAB377 | For staining mature neurons |
| Olympus VS200 slide scanner and VSI software. | Olympus Life Science | https://www.olympus-lifescience.com/en/downloads/detail-iframe/?0[downloads][id]=847254104 | Imaging software |
| Paraformaldehyde | University Medstore | PAR070.1 | For protein fixation (https://www.uoftmedstore.com/index.sz) |
| Perfusion pump | VWR International | https://pr.vwr.com/store/product/4787969/vwr-variable-speed-peristaltic-pumps | Needed for perfusion |
| Scalpel | University Medstore | 2580-M90-10 | For surgery (https://www.uoftmedstore.com/index.sz) |
| Stereotaxic | Provided by animal facility | N/A | So secure the animal for surgery |
| Surgical scissor | University Medstore | 22751-A9-240 | For surgery (https://www.uoftmedstore.com/index.sz) |
| Surgical tape | University Medstore | https://www.amazon.ca/3M-Micropore-Tape-1530-2-Rolls/dp/B0082A9GS2 | Secure the animal on the diaper |
| Sutures | University Medstore | 2297-VS881 | For surgery (https://www.uoftmedstore.com/index.sz) |
| X2 tweezers | University Medstore | 7731-A10-612 | For surgery (https://www.uoftmedstore.com/index.sz) |
References
- Kadry, H., Noorani, B., Cucullo, L. A blood-brain barrier overview on structure, function, impairment, and biomarkers of integrity. Fluids Barriers CNS. 17 (1), 69 (2020).
- Sethi, B., Kumar, V., Mahato, K., Coulter, D. W., Mahato, R. I. Recent advances in drug delivery and targeting to the brain. J Control Release. 350, 668-687 (2022).
- Vagnucci, A. H., Li, W. W. Alzheimer's disease and angiogenesis. Lancet. 361 (9357), 605-608 (2003).
- Banks, W. A., et al. Lipopolysaccharide-induced blood-brain barrier disruption: roles of cyclooxygenase, oxidative stress, neuroinflammation, and elements of the neurovascular unit. J Neuroinflamm. 12, 223 (2015).
- Archie, S. R., Shoyaib, A. A., Cucullo, L. Blood-brain barrier dysfunction in CNS disorders and putative therapeutic targets: an overview. Pharmaceutics. 13 (11), 1779 (2021).
- Jefferies, W. A., Price, K. A., Biron, K. E., Fenninger, F., Pfeifer, C. G., Dickstein, D. L. Adjusting the compass: new insights into the role of angiogenesis in Alzheimer's disease. Alzheimers Res Ther. 5 (6), 64 (2013).
- Kook, S. Y., Seok-Hong, H., Moon, M., Mook-Jung, I. Disruption of blood-brain barrier in Alzheimer disease pathogenesis. Tissue Barriers. 1 (2), e23993 (2013).
- Kempermann, G. Activity dependency and aging in the regulation of adult neurogenesis. Cold Spring Harb Perspect Biol. 7 (11), a018929 (2015).
- Del Brutto, V. J., Ortiz, J. G., Biller, J. Intracranial arterial dolichoectasia. Front Neurol. 8, 344 (2017).
- de la Torre, J. C., Mussivand, T. Can disturbed brain microcirculation cause Alzheimer's disease. Neurol Res. 15 (3), 146-153 (1993).
- Gutierrez, J., Sacco, R. L., Wright, C. B. Dolichoectasia-an evolving arterial disease. Nat Rev Neurol. 7 (1), 41-50 (2011).
- Desai, B. S., Schneider, J. A., Li, J. L., Carvey, P. M., Hendey, B. Evidence of angiogenic vessels in Alzheimer's disease. J Neural Transm. 116 (5), 587-597 (2009).
- Biron, K. E., Dickstein, D. L., Gopaul, R., Jefferies, W. A. Amyloid triggers extensive cerebral angiogenesis causing blood brain barrier permeability and hypervascularity in Alzheimer's disease. PLoS One. 6 (8), e23789 (2011).
- Zenaro, E., Piacentino, G., Constantin, G. The blood-brain barrier in Alzheimer's disease. Neurobiol Dis. 107, 41-56 (2017).
- Brandl, S., Reindl, M. Blood-Brain barrier breakdown in neuroinflammation: current in vitro models. Int J Mol Sci. 24 (16), 12699 (2023).
- Austin, B. P., et al. Effects of hypoperfusion in Alzheimer's disease. J Alzheimers Dis. 26, 123-133 (2011).
- Jagust, W. J., Budinger, T. F., Reed, B. R. The diagnosis of dementia with single photon emission computed tomography. Arch Neurol. 44 (3), 258-262 (1987).
- Schuff, N., et al. Cerebral blood flow in ischemic vascular dementia and Alzheimer's disease, measured by arterial spin-labeling magnetic resonance imaging. Alzheimers Dement. 5 (6), 454-462 (2009).
- Singh, C., Pfeifer, C. G., Jefferies, W. A. Pathogenic Angiogenic Mechanisms in Alzheimer's Disease. Physiologic and Pathologic Angiogenesis - Signaling Mechanisms and Targeted Therapy. , (2017).
- Dai, D., Kadirvel, R., Rezek, I., Ding, Y. H., Lingineni, R., Kallmes, D. Elastase-induced intracranial dolichoectasia model in mice. Neurosurgery. 76 (3), 337-343 (2015).
- Liu, F. X., et al. Modified protocol for establishment of intracranial arterial Dolichoectasia model by injection of elastase into cerebellomedullary cistern in mice. Front Neurol. 13, 860541 (2022).
- Lee, A. Y., Han, B., Lamm, S. D., Fierro, C. A., Han, H. Effects of elastin degradation and surrounding matrix support on artery stability. Am J Physiol Heart Circ Physiol. 302 (4), H873-H884 (2012).
- Li, Y., et al. The role of elastase in corneal epithelial barrier dysfunction caused by Pseudomonas aeruginosa exoproteins. Invest Ophthalmol Vis Sci. 62 (9), 7 (2021).
- Temesvári, P., Ábrahám, C. S., Gellén, J., Speer, C. P., Kovács, J., Megyeri, P. Elastase given intracisternally opens blood-brain barrier in newborn piglets. Biol Neonatol. 67 (1), 59-63 (1995).
- Takata, F., et al. Elevated permeability of the blood-brain barrier in mice intratracheally administered porcine pancreatic elastase. J Pharmacol Sci. 129 (1), 78-81 (2015).
- Hanyu, H. Diagnosis and treatment of mixed dementia. Brain Nerve. 64 (9), 1047-1055 (2012).
- Chui, H. C., Ramirez-Gomez, L. Clinical and imaging features of mixed alzheimer and vascular pathologies. Alzheimers Res Ther. 7 (1), 21 (2015).
- Saito, T., et al. Single app knock-in mouse models of alzheimer's disease. Nat Neurosci. 17 (5), 661-663 (2014).
- Dobrin, P. B., Canfield, T. R. Elastase, collagenase, and the biaxial elastic properties of dog carotid artery. Am J Physiol. 247, H124-H131 (1984).
- Wagenseil, J. E., Ciliberto, C. H., Knutsen, R. H., Levy, M. A., Kovacs, A., Mecham, R. P. Reduced vessel elasticity alters cardiovascular structure and function in newborn mice. Circ Res. 104 (10), 1217-1224 (2009).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved