A subscription to JoVE is required to view this content. Sign in or start your free trial.
Integrated One-Pot Process to Fabricate and Impregnate Starch Aerogels in Supercritical Carbon Dioxide
Not Published
In This Article
Summary
This protocol describes the development of a one-pot strategy for the fabrication and impregnation of starch aerogels. Some modifications were made within the traditional fabrication process, which allowed the integration of the three critical steps (gelatinization, retrogradation, and drying) of aerogel fabrication into a single step.
Abstract
The goal of this work was to develop a one-pot strategy for the fabrication and impregnation of starch aerogels with green coffee oil (GCO) in supercritical carbon dioxide (scCO2). For that purpose, different modifications were made to the production process to improve the integration of the three essential steps of aerogel fabrication. A strategy based on supercritical extraction (SCE) was proposed to address the conventional drying process, as well as the utilization of CO2 during the aerogel fabrication steps. The development of a novel drying approach was the most challenging task of this work as it should be performed without any solvent-exchange step. The results show that aerogels with high surface area (95 m2.g-1) could be produced using a continuous flow of CO2/ethanol (20 MPa, 40 °C, 2 mL.min-1 CO2 with 11% v/v ethanol). The next step comprised the formation of aerogels in the presence of CO2. The optimal surface area was 185 m2.g-1. Finally, the integration of all the above steps was achieved, and gelatinization, retrogradation, and drying happened sequentially in the same vessel under CO2 atmosphere. This one-pot fabrication was followed by the impregnation with GCO using a high-pressure injection step. An impregnation efficiency of 24% was obtained with this one-pot strategy confirming that a fully integrated process for the fabrication and impregnation of starch aerogels could be attained.
Introduction
The fabrication of starch aerogels is often described in three major steps: formation of hydrogels, formation of alcohol gel through solvent exchange step, and drying1,2. The formation of hydrogel involves two steps: gelatinization followed by retrogradation. The gelatinization promotes irreversible physical changes3,4,5 in the structure of starch and results from the swelling and denaturation of starch granules. Retrogradation promotes the restructuring of free amylose, leading to the formation of a three-dimensional (3D) network. Drying, which is necessary to obtain an aerogel, usually includes a solvent exchange step, wherein water is progressively replaced by ethanol followed by CO2 SCE to remove the ethanol6. The solvent exchange step is vital to obtain high-quality aerogels and is usually the focus of different studies to improve the structural properties of these materials7,8.
For example, Mehling and coworkers1 have used a multistage solvent exchange process (5 days) followed by supercritical extraction (SCE) to produce aerogels of high surface area (72 and 90 m2.g-1) from potato starch. Following the same concept, Zou and Budtova9 used a multistage solvent exchange process (4 days) followed by SCE over 5 h. These authors have obtained materials of surface area between 8 and 120 m2.g-1. Furthermore, Santos-Rosales and co-workers10 have used a multistage solvent exchange (48 h between each stage) followed by SCE over 4 h. This strategy allowed the production of aerogels of high surface area (183−228 m2.g-1). Another good example of starch aerogels with a surface area of 234 m2.g-1 is from the work of García-Gonzales et al.2 who reported a solvent exchange procedure over 24 h followed by four SCEs. A similar approach was reported by Ubeyitogullari and Ciftci11 to fabricate wheat starch aerogels with a surface area of 59.7 m2.g-1.
Despite the production of improved aerogel materials in the above studies, all of them describe a complex solvent exchange method that can last between one and five days. In fact, this complex procedure is one of the major bottlenecks in aerogel production. Hence, the improvement of the drying procedure is essential to improve the whole fabrication process, which has encouraged the development of the present protocol. Therefore, the first goal of this study was to integrate the solvent exchange with the SCE in one single step. This strategy presented a clear advantage when compared with the several examples described above, as high-quality materials were obtained by SCE using a mixture of CO2 and ethanol. Only a few studies in the literature describe a similar approach12,13,14,15. For instance, Comin and co-workers12 have reported a similar approach to dry starch aerogels involving a two-stage extraction process consisting of a 4 h ethanol extraction (0.3 mL.min-1) followed by 1 h of scCO2 extraction (1 L.min-1, 15 MPa and 40 °C).
Nevertheless, the materials produced exhibited a low surface area (<10 m2.g-1) and extensive cracking. Moreover, Brown et al.11 have reported similar problems using a continuous solvent exchange process. Even though both studies produced aerogels with properties suitable for impregnation of bioactive molecules, none of these studies could produce high-quality materials integrating both solvent exchange and SCE steps. Gurikov et al.14 reported the preparation of alginate aerogels using a static high-pressure extraction with different scCO2/ethanol/water mixtures. Their results also show that the presence of scCO2 improved the solvent exchange step. More recently, Lebedev et al.15 also reported a static extraction strategy similar to that of Gurikov et al.14. These authors showed that an scCO2/2-propanol/water mixture was able to increase the surface area of alginate aerogels. These examples highlight the novelty and the success of the protocol described herein. Another goal of the present protocol was to evaluate the possibility of using CO2 to improve the properties of the aerogels. In fact, different authors have approached this subject. Francisco and Sivik16 evaluated the impact of CO2 on the gelatinization of different types of starches. Their results show that starch gelatinization could be attained at a lower temperature in the presence of scCO2. This result was explained by plasticization and hydrostatic effects of CO2 on the structure of starch.
Moreover, Muljana and co-workers17 also reported a similar effect of CO2 on the gelatinization temperature. Thus, this protocol also introduces the positive effect that CO2 has on the production process. The combination of gelatinization, retrogradation, and drying in a one-pot process not only reduces the overall complexity of aerogel fabrication, but also allows the production of high-quality materials for impregnation with bioactive molecules. Additionally, an impregnation step was also added to this procedure to demonstrate a novel concept for the development of aerogel-based products. For this purpose, GCO was used because of its beneficial impact on human health18,19,20,21. Thus, the use of this fully integrated protocol for the fabrication/impregnation of aerogels could lead to the development of different, novel nutraceutical products.
Protocol
1. Development of a supercritical drying method by using a mixture of supercritical carbon dioxide and ethanol
- Prepare a hydrogel in the traditional way by heating 10 mL of a 15% w/v starch solution in water in a beaker at 120 °C for 20 min with magnetic stirring (600 rpm), as described previously1,2,22, to promote starch gelatinization.
- For the retrogradation step, transfer 2 mL of this solution to a cylindrical tube, and incubate at 4 °C for 48 h.
- For the supercritical drying step, place the retrograded hydrogel in a 20 mL high-pressure, variable-volume cell. Transfer the cell to a thermostatic water bath at 40 °C and pressurize it with scCO2 until the desired pressure (10-20 MPa).
NOTE: Here, the cell was pressurized at 1 MPa.min-1. - Ensure a continuous flow of scCO2 (2 mL.min-1), containing ethanol (11% (v/v) or 22% (v/v), which corresponds to 0.22 or 0.44 mL.min-1), through the reactor for 4 h using a continuous high-pressure pump. Pump the ethanol into the system using a second high-performance liquid chromatography (HPLC) pump.
- After this period, stop the ethanol flow, and start the flow of scCO2 at 2 mL.min-1 through the reactor for an additional 2 h. Leave the system to depressurize at a rate of 0.3 MPa.min-1.
NOTE: A rapid depressurization process can promote fractures in the structure of the aerogel. The drying apparatus is depicted in Figure 1.
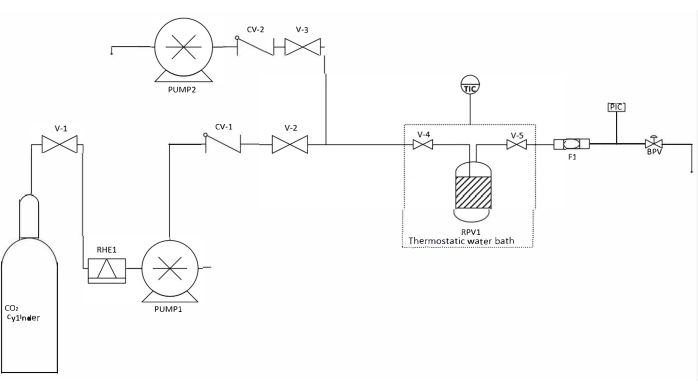
Figure 1: Schematic representation of the drying apparatus. Abbreviations: V-1-5 = valves; RHE1 = refrigerated heat exchanger; CV-1, 2 = check valves; PUMP1 = liquid CO2 pump; PUMP2 = high-performance liquid chromatography co-solvent pump; RPV1 = reactor pressure vessel; TIC = temperature indicator-controller; F1 = filter; PIC = pressure indicator-controller; BPV = Back pressure valve. Please click here to view a larger version of this figure.
2. Fabrication of starch aerogel in the presence of CO 2
NOTE: Three different temperatures (40, 90, and 120 °C) were used to evaluate the impact of CO2 on the structural properties of the aerogel.
- Dissolve 0.5 g of starch in 5 mL of water in a 10 mL high-pressure reactor. Heat the reactor to the desired temperature and then pressurize with scCO2 at 1 MPa.min-1 to a final pressure of 20 MPa. Stir the pressurized starch solution for 20 min at 600 rpm to promote gelatinization, and then cool the reactor to room temperature.
- For retrogradation, leave the pressurized starch solution in the reactor at 4 °C for 48 h. Then, place the reactor in a thermostatic water bath at 40 °C, and pressurize with scCO2 at 1 MPa.min-1 until the pressure reaches 20 MPa.
- Ensure a continuous flow of scCO2 (2 mL.min-1) containing 11% v/v/ ethanol through the reactor for 4 h. Use a continuous high-pressure pump to maintain the flow rate of CO2 at 2 mL.min-1. Pump the ethanol into the system using a second HPLC pump at 0.22 mL.min-1.
- After this period, stop the ethanol flow, and start the flow of scCO2 (2 mL.min-1) through the reactor over 2 h. Finally, leave the system to depressurize at a rate of 0.3 MPa.min-1. Characterize the materials obtained at the end of this process using the procedures described in section 5.
3. Evaluation of impregnation conditions
NOTE: The GCO extract and its characterization have been described previously23.
- For the supercritical impregnation process, place 0.1 g of the aerogel obtained at the end of the process described in section 1 in a 25 mL high-pressure, variable-volume cell. Leave this cell in a thermostatic bath at 40 °C, and pressurize with scCO2 at 1 MPa.min-1 until 10 MPa.
- Leave the aerogel for impregnation for three different periods (6, 12, and 24 h), and depressurize the system at a rate of 0.1 MPa.min-1.
NOTE: These impregnation conditions were based on previously reported procedures24. - Fill a 0.1 mL loop mounted on a 6-way high-pressure valve (Figure 2) with GCO extract using a syringe. Pressurize the loop with scCO2 (flow of 2 mL.min-1) until 30 MPa and leave for 10 min to equilibrate. Turn the valve to the injection position to start the flow of scCO2 across the loop until the pressure inside the reactor reaches 30 MPa. Leave the system in this condition for 6, 12, or 24 h.
NOTE: The GCO extract was obtained and characterized using previously reported procedures23.
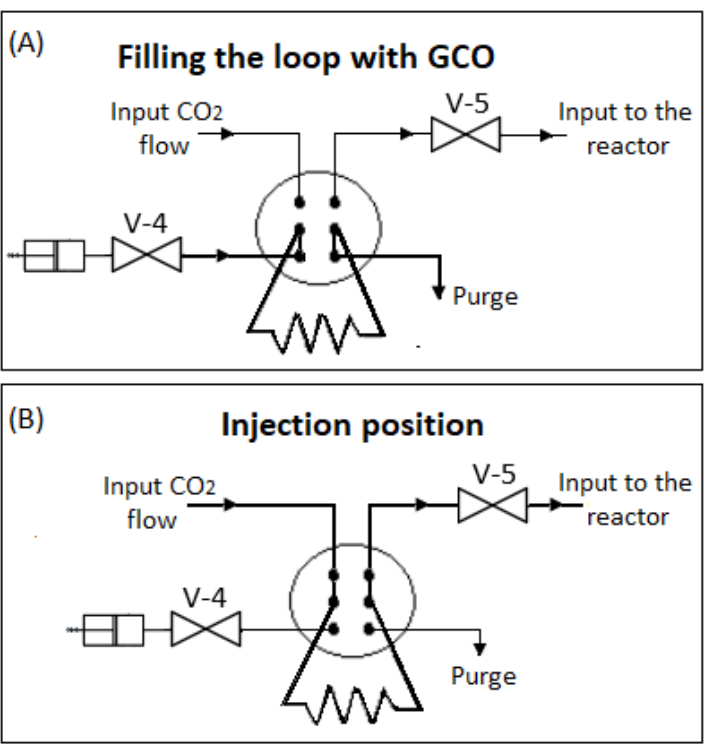
Figure 2: Schematic representation of the 6-Way high-pressure valve. (A) Filling the loop with GCO. (B) Injection position. Abbreviations: GCO = green coffee oil; V-4, 5 = valves. Please click here to view a larger version of this figure.
- After this period, depressurize the system at 0.1 MPa.min-1, and collect the impregnated material. Determine the amount of impregnated oil using the extraction procedure described in the literature1, 7, 11.
- Disperse 0.1 g of the impregnated aerogel in 5 mL of chloroform, and perform the extraction for 1 h at 50 °C with magnetic stirring (600 rpm). Filter the extracts using a 0.2 µm syringe filter, and collect the extracts in a 5 mL calibrated round flask. Evaporate the solvent under a flow of nitrogen, and determine the impregnation efficiency in mg of oil per 100 mg of aerogel.
4. One-pot process for fabrication and impregnation of starch aerogels
NOTE: This process used the optimized conditions from the previous steps using previously reported procedures24.
- To fabricate the hydrogel in the presence of CO2, dissolve 0.5 g of starch in 5 mL of water in a 10 mL high-pressure reactor. Heat the reactor to the desired temperature, and then pressurize gently with scCO2 at 1 MPa.min-1 to a pressure of 20 MPa. Stir the pressurized starch solution for 20 min at 600 rpm to promote the gelatinization, and then cool the reactor to room temperature.
- For retrogradation, leave the pressurized starch solution at 4 °C for 48 h. Then, place the reactor in a thermostatic water bath at 40 °C, and pressurize with scCO2 at 1 MPa.min-1 to a pressure of 20 MPa.
- Continuously flow scCO2 containing 11% v/v ethanol at 2 mL.min-1 through the reactor for 4 h. Use a continuous high-pressure pump to maintain the flow rate of CO2 at 2 mL.min-1 and a second HPLC pump for the ethanol at 0.22 mL.min-1.
- After this period, stop the ethanol flow, and start the flow of scCO2 at 2 mL.min-1 through the reactor for 2 h. Decrease the pressure to 10 MPa to allow the supercritical impregnation take place, and maintain the reactor temperature at 40 °C.
- Fill a 0.5 mL loop mounted on a 6-way high-pressure valve (Figure 2) with GCO extract using a syringe. Pressurize the loop with scCO2 to a achieve a pressure of 30 MPa and leave for 10 min to equilibrate. Turn the valve to the injection position, and start the flow of scCO2 across the loop until the pressure inside the reactor reaches 30 MPa; leave the system in this condition for 12 h.
NOTE: The GCO extract was obtained and characterized using previously reported procedures23. - Depressurize the system at a rate of 0.1 MPa.min-1, and collect the impregnated material. See Figure 3 for a schematic representation of the one-pot system. Determine the amount of the impregnated oil using the extraction procedure described in the literature1,7,11.
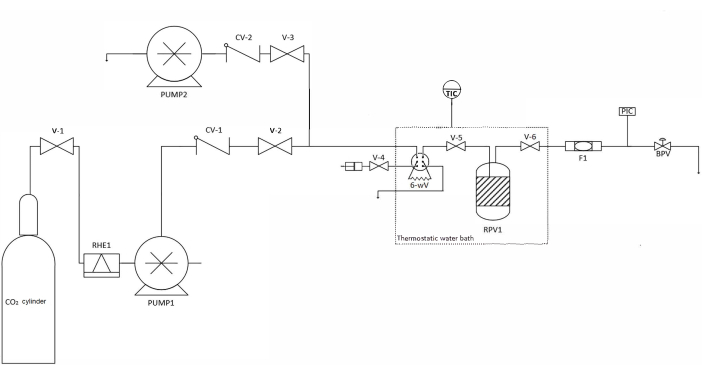
Figure 3: Schematic representation of the one-pot system. Abbreviations: V-1-6 = valves; RHE1 = refrigerated heat exchanger; CV-1, 2: check valves; PUMP1 = liquid CO2 pump; PUMP2 = high-performance liquid chromatography co-solvent pump; RPV1 = reactor pressure vessel; 6-wV = 6-way valve; TIC = temperature indicator-controller; F1 = filter; PIC = pressure indicator-controller; BPV: back pressure valve. Please click here to view a larger version of this figure.
- Disperse 0.5 g of the impregnated aerogel in 25 mL of chloroform, and stir for 1 h at 50 °C and 600 rpm for extraction. Use a 0.2 µm syringe filter to filter the extracts, collect them in a calibrated round flask, and evaporate the solvent under a flow of nitrogen. Calculate the extracted mass by determining the difference between the flask weights, and determine the impregnation efficiency in terms of mg of oil per 100 mg of aerogel.
5. Characterization of aerogels
- Determine the surface area of the aerogels using an ultra-high-purity nitrogen adsorption and desorption isotherms at 77 K using the Brunauer-Emmett-Teller (BET) method.
- Prior to the analysis, heat ~200 mg of sample at 115 °C for 4 h under vacuum.
- Evaluate adsorption isotherms in the linear region of the BET plot (at a relative pressure p/p0 in the range of 0.05-0.3) using a multipoint BET for the determination of the surface area.
- Estimate both volume and pore size using the Barrett-Joyner-Halenda (BJH) method.
- Characterize aerogel morphology using Field Emission Scanning Electron Microscopy at 5 kV and 15 mA under high vacuum. Cut the materials transversely to obtain 1 mm thickness, place the slices on a double-sided carbon film, and sputter-coat with gold22.
- Determine the diffraction pattern and the degree of crystallinity by X-ray diffraction.
- Before the measurements, crush the samples, and scatter them in the equipment holder.
Make sure the system is equipped with a copper target X-ray tube set to 40 kV and 40 mA. Expose the samples to the X-ray beam, and scan from 5 to 40 Å with an angular scanning velocity of 1.267°.min-1,17,22. - Finally, calculate the degree of crystallinity (DC) using the following equation.
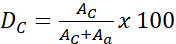 (1)
(1)
Where AC is the area under the curve for crystalline reflection, and Aa is the area under the curve for crystalline amorphous17,25.
- Before the measurements, crush the samples, and scatter them in the equipment holder.
Results
Traditionally, the process used to transform hydrogels into aerogels involves two major steps: solvent exchange and SCE. The first goal of this protocol was to integrate both steps into a continuous flow process. To that end, the impact of pressure and co-solvent (ethanol) composition on the surface area of the materials was examined. Surface area is an excellent indicator of the quality of the aerogel and allows a direct comparison with literature.
Initial studies demonstrated that the extrac...
Discussion
One of the critical steps of this protocol is the gradual removal of water in the supercritical drying step. To obtain materials with a good surface area, the flow should be maintained between 2 and 3 mL.min-1, otherwise an extensive pore-collapsed material will be obtained. Thus, judicious control of the extraction conditions is essential to obtain materials with the desirable properties for impregnation. Moreover, the size of the hydrogel is also a critical point. Large hydrogels could be difficult to dry an...
Disclosures
The authors have nothing to disclose.
Acknowledgements
We acknowledge financial support from FAPESP through the project 2015/14905-0 and to FAPESP and SHELL Brazil through the 'Research Centre for Gas Innovation - RCGI' (FAPESP Proc. 2014/50279-4), hosted by the University of Sao Paulo, and the support given by ANP (Brazil's National Oil, Natural Gas and Biofuels Agency) through the R&D levy regulation as well as The Research Center for Gas Innovation (RCGI). Maria Villegas acknowledges CAPES for her Msc grant and RCGI for her technical research position.
Materials
| Name | Company | Catalog Number | Comments |
| Equipment | |||
| 6-Way high pressure valve | Rheodyne | ||
| Field Emission Scanning Electron Microscopy | JOEL | JSM-7401 F | |
| High pressure cell 10 mL | Citua (Campinas, Brazil) | Custom made | Set Up construction done by Citua (Campinas, Brazil) |
| High pressure cell 20 mL | Citua (Campinas, Brazil) | Custom made | Set Up construction done by Citua (Campinas, Brazil) |
| High pressure p-series Pump | Thar Technologies | Model Thar SFC, P-50A | |
| HPLC-type pump | Shimadzu | Model LC-10AD Vp | |
| Polypropylene molds (eppendorf) | Eppendorf, Brazil | Safe-Lock Tube 2.0 mL | |
| Surface Area Analyzer | Quantachrome Instruments | Model Nova 1220 | |
| X-Ray Diffraction Instrument | Rigaku | Model Miniflex | Powder X-Ray Diffraction (XRD) Instrument |
| Chemicals | |||
| Ethanol anhydrous, ≥99.5% | Sigma-Aldrich, Brazil | CAS # 64-17-5 | |
| Liquid carbon dioxide, 99.98% | Oxilumen, Brazil | CAS # 124-38-9 | |
| Native potato starch | Vetec, Brazil | CAS # 9005-25-8 |
References
- Mehling, T., Smirnova, I., Guenther, U., Neubert, R. H. H. Polysaccharide-based aerogels as drug carriers. Journal of Non-Crystalline Solids. 355 (50-51), 2472-2479 (2009).
- García-González, C. A., Camino-Rey, M. C., Alnaief, M., Zetzl, C., Smirnova, I. Supercritical drying of aerogels using CO 2: Effect of extraction time on the end material textural properties. Journal of Supercritical Fluids. 66, 297-306 (2012).
- Ring, S. G., et al. The gelation and crystallisation of amylopectin. Carbohydrate Research. 162 (2), 277-293 (1987).
- Debet, M. R., Gidley, M. J. Why do gelatinized starch granules not dissolve completely? Roles for amylose, protein, and lipid in granule "ghost" integrity. Journal of Agricultural and Food Chemistry. 55 (12), 4752-4760 (2007).
- Le Corre, D., Bras, J., Dufresne, A. Starch Nanoparticles: A Review. Biomacromolecules. 11 (5), 1139-1153 (2010).
- García-González, C. A., Alnaief, M., Smirnova, I. Polysaccharide-based aerogels - Promising biodegradable carriers for drug delivery systems. Carbohydrate Polymers. 86 (4), 1425-1438 (2011).
- De Marco, I., Reverchon, E. Starch aerogel loaded with poorly water-soluble vitamins through supercritical CO2 adsorption. Chemical Engineering Research and Design. 119, 221-230 (2017).
- Druel, L., Bardl, R., Vorwerg, W., Budtova, T. Starch Aerogels: A Member of the Family of Thermal Superinsulating Materials. Biomacromolecules. 18 (12), 4232-4239 (2017).
- Zou, F., Budtova, T. Tailoring the morphology and properties of starch aerogels and cryogels via starch source and process parameter. Carbohydrate Polymers. , (2020).
- Santos-Rosales, V., et al. Stability Studies of Starch Aerogel Formulations for Biomedical Applications. Biomacromolecules. , (2020).
- Ubeyitogullari, A., Ciftci, O. N. Phytosterol nanoparticles with reduced crystallinity generated using nanoporous starch aerogels. Roysal Society of Chemistry Advances. 6 (110), 108319-108327 (2016).
- Comin, L. M., Temelli, F., Saldaña, M. D. A. Barley beta-glucan aerogels via supercritical CO 2 drying. FRIN. 48 (2), 442-448 (2012).
- Brown, Z. K., Fryer, P. J., Norton, I. T., Bridson, R. H. Drying of agar gels using supercritical carbon dioxide. The Journal of Supercritical Fluids. 54 (1), 89-95 (2010).
- Gurikov, P., Raman, S. P., Weinrich, D., Fricke, M., Smirnova, I. A novel approach to alginate aerogels: carbon dioxide induced gelation. RSC Advances. 5 (11), 7812-7818 (2015).
- Lebedev, A., Suslova, E., Troyankin, A., Lovskaya, D. Investigation of Aerogel Production Processes: Solvent Exchange under High Pressure Combined with Supercritical Drying in One Apparatus. Gels. 7 (1), (2021).
- Francisco, J. D. C., Sivik, B. Gelatinization of cassava, potato and wheat starches in supercritical carbon dioxide. Journal of Supercritical Fluids. 22 (3), 247-254 (2002).
- Muljana, H., Picchioni, F., Heeres, H. J., Janssen, L. P. B. M. Supercritical carbon dioxide (scCO2) induced gelatinization of potato starch. Carbohydrate Polymers. 78 (3), 511-519 (2009).
- Cavin, C., Mace, K., Offord, E. A., Schilter, B. Protective effects of coffee diterpenes against aflatoxin B1-induced genotoxicity: Mechanisms in rat and human cells. Food and Chemical Toxicology. 39 (6), 549-556 (2001).
- Cavin, C., Holzhaeuser, D., Scharf, G., Constable, A., Huber, W., Schilter, B. Cafestol and kahweol, two coffee specific diterpenes with anticarcinogenic activity. Food and Chemical Toxicology. 40 (8), 1155-1163 (2002).
- Lee, K. J., Choi, J. H., Jeong, H. G. Hepatoprotective and antioxidant effects of the coffee diterpenes kahweol and cafestol on carbon tetrachloride-induced liver damage in mice. Food and Chemical Toxicology. 45 (11), 2118-2125 (2007).
- Lee, K. J., Jeong, H. G. Protective effects of kahweol and cafestol against hydrogen peroxide-induced oxidative stress and DNA damage. Toxicology Letters. 173 (2), 80-87 (2007).
- Ubeyitogullari, A., Ciftci, O. N. Formation of nanoporous aerogels from wheat starch. Carbohydrate Polymers. 147, 125-132 (2016).
- De Oliveira, P. M. A., De Almeida, R. H., De Oliveira, N. A., Bostyn, S., Gonçalves, C. B., De Oliveira, A. L. Enrichment of diterpenes in green coffee oil using supercritical fluid extraction - Characterization and comparison with green coffee oil from pressing. Journal of Supercritical Fluids. 95, 137-145 (2014).
- Villegas, M., Oliveira, A. L., Bazito, R. C., Vidinha, P. Development of an integrated one-pot process for the production and impregnation of starch aerogels in supercritical carbon dioxide. Journal of Supercritical Fluids. 154, 104592 (2019).
- Nara, S., Komiya, T. Studies on the Relationship Between Water-satured State and Crystallinity by the Diffraction Method for Moistened Potato Starch. Starch - Stärke. 35 (12), 407-410 (1983).
- Cooke, D., Gidley, M. J. Loss of crystalline and molecular order during starch gelatinisation: origin of the enthalpic transition. Carbohydrate Research. 227, 103-112 (1992).
- Ratnayake, W. S., Jackson, D. S. A new insight into the gelatinization process of native starches. Carbohydrate Polymers. 67 (4), 511-529 (2007).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionExplore More Articles
This article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved