A subscription to JoVE is required to view this content. Sign in or start your free trial.
Quantification of Absolute Myocardial Flow and Resistance by Continuous Thermodilution in Patients with Ischemia and Nonobstructive Coronary Artery Disease (INOCA)
In This Article
Summary
Presented here is a protocol to measure absolute myocardial flow and resistance using continuous thermodilution in patients with ischemia and nonobstructive coronary artery disease.
Abstract
In approximately half of the patients undergoing coronary angiography for angina pectoris or for signs or symptoms suggestive of ischemic heart disease, no obstructive coronary artery disease is angiographically visible. The majority of these patients with angina or ischemia and no obstructive coronary artery disease (INOCA) have an underlying coronary vasomotor dysfunction, and current consensus documents recommend diagnostic invasive coronary vasomotor function testing (CFT).
During CFT, a variety of vasomotor dysfunction endotypes can be assessed, including vasospastic coronary dysfunction (epicardial or microvascular vasospasm), and/or microvascular vasodilatory dysfunction, including impaired vasodilatory capacity and increased microvascular resistance. The quantification of the continuous thermodilution derived absolute coronary blood flow and resistance might be a better measure compared to the currently used standard physiologic measures. This article provides an overview of this continuous thermodilution method.
Introduction
In approximately half of the patients undergoing coronary angiography for angina pectoris or for signs or symptoms suggestive of ischemic heart disease, no obstructive coronary artery disease is angiographically visible1. The majority of these patients with angina or ischemia and no obstructive coronary artery disease (INOCA) have an underlying coronary vasomotor dysfunction, and current ESC guidelines and a recent ESC position paper on INOCA recommend diagnostic invasive coronary vasomotor function testing (CFT)1,2.
During CFT, a variety of vasomotor dysfunction endotypes can be assessed, including vasospastic coronary dysfunction (epicardial or microvascular vasospasm), and/or microvascular vasodilatory dysfunction, including impaired vasodilatory capacity and increased microvascular resistance. Consensus criteria for these endotypes have been defined by the Coronary Vasomotion Disorders International Study Group (COVADIS)3,4.
While vasospastic coronary dysfunction is generally demonstrated by acetylcholine provocation testing, the diagnosis of microvascular vasodilatory dysfunction is more complex. This diagnosis is made by an abnormal index of microvascular resistance (IMR) and/or coronary flow reserve (CFR)4.
Two methods exist for the measurement of IMR or CFR: thermodilution or Doppler flow velocity. Both use intravenous adenosine to induce maximal hyperemia (and thus minimal resistance), and both methods have been extensively validated. However, they do have several important shortcomings: the need for adenosine limits their use in patients with severe chronic obstructive pulmonary disease or asthma. Also, the thermodilution method may overestimate the CFR and has a large intra-observer variability, and with the Doppler flow velocity methods it can be challenging to obtain a stable Doppler flow signal5. Most importantly, both CFR and IMR are only surrogate measures and fail to quantify true coronary blood flow and resistance.
Absolute coronary blood flow (Q) and resistance (R) can be directly quantified with the use of a recently validated and novel method that uses continuous thermodilution with intracoronary saline infusion at room-temperature to induce hyperemia. A dedicated monorail infusion catheter and a pressure wire with temperature sensors enables direct quantification of Q and R, without the use of adenosine. This novel method has been shown to be safe, highly reproducible and operator-independent 6,7.
As has been urged by a recent consensus statement, we need a better understanding of the underlying mechanism of myocardial ischemia in patients with INOCA, across the different endotypes1. This could have major implications for treatment and prognosis. The quantification of the absolute coronary blood flow and resistance might be a better measure compared to the currently used standard physiologic measures. It was recently shown that continuous thermodilution measurements are associated with symptoms in INOCA, while IMR and CFR were not8. Additional outcome data will follow. In this article, the continuous thermodilution protocol is described.
Protocol
The following protocol was approved by the local medical ethics committee at the Radboudumc hospital, Nijmegen, the Netherlands. The following steps should be followed when performing continuous thermodilution to calculate absolute flow and resistance.
1. Preparations
- Withhold vasoactive medications for at least 24 hours (48 hours in case of calcium channel blockers).
2. Diagnostic Coronary Angiography
- Inject local anesthesia (1 to 2 mL of 20 mg/mL lidocaine) either in proximity to the right radial artery or the left radial artery. In case radial access is not possible, inject local anesthesia either in proximity to the right femoral artery (usually 10 mL of lidocaine) or the left femoral artery.
- Confirm local anesthesia by pricking the anesthetized-skin with the needle and check whether pain is still present.
- Puncture the radial or femoral artery with a cannula, insert the wire through the cannula and remove it. Insert a 6 Fr sheath over the wire. Make sure to perform this under sterile conditions.
- Administer heparin according to local protocol (patient weight-adjusted 100 international units per kilogram, minimum of 5000 international units, maximum of 10.000 international units).
- Advance the wire through the sheath to the ascending aorta and place the diagnostic catheter above the aortic valve. Then remove the wire and connect the catheter with the contrast syringe.
- Engage the right coronary artery with a diagnostic catheter. After intracoronary administration of 0.2 mg of nitroglycerin, perform coronary angiography with manual injections of contrast agent.
- Engage the left coronary artery with a guiding catheter (to avoid switching from diagnostic to guiding afterwards). After intracoronary administration of 0.2 mg of nitroglycerin, perform coronary angiography with manual injections of a contrast agent. Use a 6 Fr size guiding catheter or larger to facilitate the dedicated monorail catheter (step 4).
- Exclude the presence of obstructive coronary artery disease: any relevant epicardial stenosis by visual assessment, and intracoronary physiological assessment in case of an intermediate epicardial stenosis (40-90% angiographic stenosis) 9.
3. Set up of continuous thermodilution measurements
- Ensure that all coronary pressure tracings and temperatures are wirelessly transmitted and analyzed by a dedicated console equipped with software that automatically calculates Q and R.
- Prepare a contrast injector with pressure-limit containing 100 to 150 mL of saline at room temperature.
4. Continuous thermodilution measurements
- Manually flush a guidewire with pressure and temperature sensors (further referred to as the "pressure wire") using syringes with saline.
- Make sure the pressure wire is connected to the appropriate software (so that pressure/temperature are visualized real-time) and pass the pressure wire through the guiding catheter. Ensure that the proximal part of the radiopaque section of the pressure wire is placed in the ostium of the coronary artery using angiography.
- Equalize the pressure wire to the aortic pressure. It is essential that this step is completed before any measurements are initiated.
- Advance the pressure wire to the distal 1/3 of the coronary artery of interest.
- Connect a dedicated monorail catheter to the contrast injector.
- Flush the monorail catheter with saline using the automated contrast injector set at 10 mL/min to remove air from the monorail catheter. When saline exits the infusion holes at the tip of the monorail catheter, advance the monorail catheter through the guiding catheter while flushing.
- When the monorail catheter passes the hemostatic connector, stop flushing and advance the monorail catheter further into the guiding catheter.
- Place the radiopaque marker of the monorail catheter in the proximal part (first centimeter) of the coronary artery of interest.
NOTE: Make sure there is at least 3 cm distance between the radiopaque marker of the proximally placed monorail catheter and the radiopaque marker of the distally placed guidewire to ensure optimal mixing of blood and saline 10,11. - On the dedicated software, select the Absolute flow program, select the coronary artery of interest, and set the temperature to zero (reference temperature comparable to 37 °C in humans).
- Select the appropriate infusion rate of (room-temperature) saline (Qi) on the infusion pump and make sure the program settings are updated accordingly.
NOTE: Qi is usually 20 mL/min for the left anterior descending (LAD) and left circumflex artery (LCX) and 15 mL/min for the right coronary artery (RCA). Do not start the infusion yet. - Start the absolute flow measurement on the dedicated software program. Do not stop the measurement until step 4.16. Ensure several seconds are measured before the infusion of saline is initiated.
- Start the infusion of saline at the infusion pump at the selected flow rate (Qi). Do not stop infusion until step 4.15.
NOTE: After the start of the infusion with saline, the temperature in the distal coronary artery will decrease. - Ensure that a steady state distal temperature of the blood/saline mixture is recorded for at least 10 s.
- Pull the guidewire back into the monorail infusion catheter to obtain a proximal temperature (Ti). Ensure that a stable Ti is recorded for approximately 10 s.
- Stop the infusion of room temperature saline on the infusion pump.
- Ensure the temperature returns to the zeroed value. After this, stop the absolute flow measurement on the dedicated software program.
NOTE: FFR, absolute Q and R are now automatically calculated by the dedicated software. - Remove the dedicated monorail catheter.
- Ensure that the pressure wire is placed in the ostium of the coronary artery similar to step 4.2 and perform a drift check of the pressure measurement. If drift is more than 2 mmHg, repeat measurements. If there is no drift, remove the pressure wire.
- Perform one or two final angiographies visualizing the coronary artery of interest to check for possible complications (e.g., coronary artery dissection).
5. Calculation of absolute flow and absolute resistance
NOTE: As shown in Figure 1, the dedicated monorail infusion catheter allows the infusion of saline only through four outer side holes, resulting in complete and optimal mixing with blood; two inner side holes allow the measurement of temperature by the used guidewire.
- Calculate absolute flow (Q) in mL per minute with the following equation7:
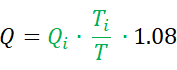
As mentioned above, Qi is the saline infusion rate in mL per minute and Ti is the temperature of the infused saline near the dedicated monorail catheter exit. T is the temperature of the homogenous mixture of blood and saline in the distal part of the coronary artery during infusion. The constant 1.08 relates to the difference between the specific temperature and density of blood and saline, and when saline is infused in blood. - Calculate R, expressed in Wood Units (WU) or mmHg * (L/min), by the formula:
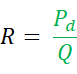
There is a readily available advanced platform to measure physiological indices, that communicates with the pressure wire and that allows live calculation of FFR, Q and R (Table of Materials).
Results
Figure 2 shows a representative measurement performed in patient A with no obstructive CAD on coronary angiography. The LAD artery was measured using continuous thermodilution to calculate absolute Q and R. The red and green lines represent pressure measurements, and the blue line represents the temperature curve. The infusion rate was set at 20 mL/min (Qi) since the LAD artery was measured. At point 1, the infusion was started and the temperature measured at the distally placed pressur...
Discussion
Continuous thermodilution is an accurate method to measure absolute coronary flow and resistance, which has been shown to strongly agree with the gold standard [15O2]H2O PET derived flow and resistance5. These measurements are of special interest in INOCA patients, with current clinical guidelines recommending the assessment of coronary flow and resistance in this group.
Fractional flow reserve (FFR), the ratio of the maximal myocardial ...
Disclosures
Peter Damman received lecture and/or consultancy fees from Phillips and Abbott Vascular.
Acknowledgements
None.
Materials
| Name | Company | Catalog Number | Comments |
| Rayflow multipurpose infusion catheter | Hexacath | RFW61S | Only compatible with 6F guiding catheter |
| PressureWire X guidewire | Abbott | C12059 | Wireless guidewire with distal temperature and pressure sensor |
| Coroventis CoroFlow Cardiovascular System software | Coroventis | N/A | Advanced platform to measure physiological indices |
| Illumena Neo injector or similar injector system | Liebel-Flarsheim | GU01181006-E | Any injector with pressure limit (600 psi) and adjustable flow and volume injection rate |
| 100 ml NaCl 0.9% at room temperature |
References
- Kunadian, V., et al. An EAPCI Expert Consensus Document on Ischaemia with Non-Obstructive Coronary Arteries in Collaboration with European Society of Cardiology Working Group on Coronary Pathophysiology & Microcirculation Endorsed by Coronary Vasomotor Disorders International Study Group. European Heart Journal. 41 (37), 3504-3520 (2020).
- Knuuti, J., et al. ESC Guidelines for the diagnosis and management of chronic coronary syndromes. European Heart Journal. 41 (3), 407-477 (2020).
- Beltrame, J. F., et al. International standardization of diagnostic criteria for vasospastic angina. European Heart Journal. 38 (33), 2565-2568 (2017).
- Ong, P., et al. International standardization of diagnostic criteria for microvascular angina. International Journal of Cardiology. 250, 16-20 (2018).
- Everaars, H., et al. Continuous thermodilution to assess absolute flow and microvascular resistance: validation in humans using [15O]H2O positron emission tomography. European Heart Journal. 40 (28), 2350-2359 (2019).
- Konstantinou, K., et al. Absolute microvascular resistance by continuous thermodilution predicts microvascular dysfunction after ST-elevation myocardial infarction. International Journal of Cardiology. 319, 7-13 (2020).
- Xaplanteris, P., et al. Catheter-Based Measurements of Absolute Coronary Blood Flow and Microvascular Resistance: Feasibility, Safety, and Reproducibility in Humans. Circulation Cardiovascular Interventions. 11 (3), 006194 (2018).
- Konst, R. E., et al. Absolute Coronary Blood Flow Measured by Continuous Thermodilution in Patients With Ischemia and Nonobstructive Disease. Journal of the American College of Cardiology. 77 (6), 728-741 (2021).
- Neumann, F. J., et al. ESC/EACTS Guidelines on myocardial revascularization. European Heart Journal. 40 (2), 87-165 (2019).
- Aarnoudse, W., et al. Direct volumetric blood flow measurement in coronary arteries by thermodilution. Journalof the American College of Cardiology. 50 (24), 2294-2304 (2007).
- van't Veer, M., et al. Novel monorail infusion catheter for volumetric coronary blood flow measurement in humans: in vitro validation. EuroIntervention: Journal of EuroPCR in collaboration with the Working Group on Interventional Cardiology of the European Society of Cardiology. 12 (6), 701-707 (2016).
- Fournier, S., et al. Normal Values of Thermodilution-Derived Absolute Coronary Blood Flow and Microvascular Resistance in Humans. EuroIntervention: Journal of EuroPCR in collaboration with the Working Group on Interventional Cardiology of the European Society of Cardiology. , (2020).
- Keulards, D. C. J., et al. Safety of Absolute Coronary Flow And Microvascular Resistance Measurements by Thermodilution. EuroIntervention: Journal of EuroPCR in collaboration with the Working Group on Interventional Cardiology of the European Society of Cardiology. , (2020).
- Konijnenberg, L. S. F., et al. Pathophysiology and diagnosis of coronary microvascular dysfunction in ST-elevation myocardial infarction. Cardiovascular Research. 116 (4), 787-805 (2020).
- Wijnbergen, I., van't Veer, M., Lammers, J., Ubachs, J., Pijls, N. H. Absolute coronary blood flow measurement and microvascular resistance in ST-elevation myocardial infarction in the acute and subacute phase. Cardiovascular Revascularization Medicine: including Molecular Interventions. 17 (2), 81-87 (2016).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved