A subscription to JoVE is required to view this content. Sign in or start your free trial.
Unfixed Head and Closed Skull Weight Drop Method as a Rodent Model of Mild Traumatic Brain Injury (Mtbi) for Studies of Post-Traumatic Headache (PTH)
Not Published
In This Article
Summary
The protocol describes a rodent model of unfixed head mild traumatic brain injury induced by a weight drop allowing for free linear and rotational impact-related forces. The dynamics of this injury are similar to concussions that occur in humans and are useful to investigate underlying mechanisms involved in post-traumatic headache.
Abstract
Mild traumatic brain injury (mTBI) represents one of the major contributors to the overall global disability. Post-traumatic headache (PTH) is a frequent and often the most debilitating associated symptom. For some patients, headache resolves in the first weeks to 3 months following a mTBI, a condition defined as acute PTH (APTH). However, several individuals continue to experience ongoing or intermittent headaches that persist beyond 3 months, a condition defined as persistent PTH (PPTH). Many animal models of TBI have been developed. Often, these are performed by impacting the head of rodents whose motion is mechanically fixed and/or involves an open skull. The following protocol describes a model where the head is unfixed, allowing free linear and rotational forces induced by a standardized weight drop that has similarities to concussions that occur in many situations in humans. The headache in this model is characterized by initial transient tactile allodynia in the cephalic region that lasts for several days and reflects the pain-like feature of APTH. Tactile allodynia is also observed in the extra-cephalic body areas suggestive of central sensitization and PTH. Following resolution of the transient APTH, animals display long-lasting latent sensitization, where baseline pain responses are normal, but otherwise innocuous provocative stimuli provoke cephalic and hind paw tactile allodynia, suggesting the development of persistent sensitization that may underlie PPTH. The key advantages of this model include concussion-like injury dynamics (closed head injury, reproduction of both linear and rotational forces) and induction of transient pain-like features followed by a long-lasting (persistent) period of increased vulnerability to normally innocuous stimuli that may reinstate a headache-like response. This method may be used to investigate the underlying pathological mechanisms involved in the transition of APTH to PPTH and the evaluation of novel therapeutic strategies for the treatment or prevention of APTH and PPTH.
Introduction
Mild traumatic brain injury (mTBI), also known as concussion, represents the most prevalent type of closed head injury and is one of the leading causes of global disability1,2,3,4,5. mTBI often affects civilians, military personnel, and veterans and can result from blunt trauma, acceleration, or deceleration forces, including falls, motor vehicle collisions, contact or collision sports, exposure to blasts, and additional mechanisms of injury1,2. Post-traumatic headache (PTH) is the most common and often incapacitating symptom following mTBI. Headache attributed to mTBI that resolves within 3 months is considered as acute PTH (APTH), while headache symptoms that persist beyond 3 months and can last for years are defined as persistent PTH (PPTH)1,2,3. Approximately 50% of individuals who develop APTH after mTBI will progress to PPTH6.
In spite of significant advances in clinical studies of PTH, there is still no approved therapy for this condition1,2,7. The pathophysiological changes underlying PTH and/or those driving transition to PPTH remain poorly understood. Multiple mechanisms have been suggested to contribute to PTH/PPTH, including genetic phenotype, prior existing conditions such as migraine, neuroplasticity of descending pain modulatory pathways, brain structural and functional remodeling, and others8. PTH most often presents with a migraine phenotype, suggesting overlapping pathophysiological mechanisms, including the likely involvement of calcitonin-gene-related peptide (CGRP) signaling1,7. Nevertheless, clinical reports suggest that most migraine therapeutics, including those modulating CGRP are not very effective for PTH9. Further studies are warranted for a better understanding of pathophysiological mechanisms associated with PTH in order to discover improved therapies to control PTH and to prevent the progression to PPTH.
Animal models of mTBI ideally should closely consider clinical symptoms in order to increase potential mechanistic translation. Recently, models of TBI have been classified into three main categories based on the method of induction. These methods include weight drop, piston-driven impacts, and "other" models that are described as "unconventional"10. The weight drop models constitute the largest group and consist of dropping a projectile with specific characteristics (material, size, weight) through a guide tube placed at a specific height directly onto the head of the animal10. Extensive variations of the weight drop method to induce mTBI in rodents can be found in the literature10. Piston-driven models of mTBI, such as controlled cortical impact (CCI), consist of a piston that produces injury related to a specific depth, velocity, and/or impact force when considering the surface of the skull or scalp as the zero or the hit mark10. Impact devices can include pistons driven by compressed nitrogen, electromagnetic or pneumatic mechanisms10. In addition, penetrating injuries may also be induced by a pulse of pressurized fluid (i.e., fluid percussion, FPI) through a craniotomy7. However, while the exact method and the severity of injury induced by CCI and FPI can be varied, they often produce more severe injuries with substantial brain tissue loss and neuropathological outcomes that may better resemble severe TBI versus the mild TBI concussive injuries most commonly associated with PTH and PPTH in humans7. Most of the studies using the weight drop and the piston model of mTBI have used adult mice and rats with additional variables that included adaptations according to the species, strain, and size of the rodent10. Many other types of mTBI models have also been described in the literature, including a spring-loaded launching, the projectile model, a rubber reflex hammer fixed into a pivot, a microTBI surgical technique, and a model of fall, which induces mTBI by dropping the animals onto an immovable surface10.
Our group has adapted a weight drop model of mTBI allowing free rotational and linear head movements, previously described by Khun and colleagues11,12,13,14. The weight drop apparatus used to mimic mTBI induction reported in this protocol is represented in Figure 1. It consists of a vertical guide metal tube (124 cm) that holds and guides the weight that is dropped onto the head of a lightly anesthetized mouse. A PMMA (Poly(methyl methacrylate)) box (20 cm x 20 cm x 16.5 cm) with an opening on the top (12 cm x 8 cm) allows the placement of the animal and allows the linear and rotational movement of the head after the impact from the weight drop. The bottom of the box contains one 2.5 cm-thick wavy and two 1.25 cm-thick flat acoustic foam cushions (15 cm x 18 cm) to support and comfort the animal after the impact from the weight drop. The cushion and the bottom of the PMMA box also have an opening through which the weight goes, preventing a second hit during the fall of the animal. The opening of the top of the PMMA box is covered by a single layer of tissue paper, with perforation (11 cm x 12 cm) with the ends taped, which supports the body weight of a mouse up to 50 g for mTBI impact or sham procedures (no impact). The thinness of the paper stage produces little or no resistance upon the weight drop impact. The projectile to induce the weight drop (20 cm and 0.3 cm diameter) is made of steel, weighs 100 g, and has a flat end with radiused edges (0.3 cm) to restrict the zone of contact to the top of the head between the ears and the eyes of the mouse. The weight is released from a height of 37 inches above the head. The weight drop impacts the sagittal suture midway between bregma and lambda. The weight is placed on the top of the guide tube, with the flat end with radiused edges pointing down toward the animal, and is held in place by an L-shaped steel trigger (5 cm x 2 cm). The weight is dropped vertically through the guide tube by removing the trigger.

Figure 1: The weight drop apparatus for mTBI induction to the unfixed head of a mouse. (A) The apparatus consists of a vertical guide metal tube (124 cm) for the weight drop and a PMMA box (20 cm x 20 cm x 16.5 cm) with a 5 cm acoustic foam cushion in the bottom (one 2.5 cm thick wavy acoustic foam and two 1.25 cm thick flat acoustic foam) (B). The 100 g steel weight with (C) dimensions (20 cm and 0.3 cm diameter) has a flat end with radiused edges (0.3 cm), which is placed on the top of the guide tube and is held in place by a steel trigger (red circle on panel A). The weight is released from a height of 94 cm above the head. (D) The top of the PMMA box has a 12 cm x 8 cm opening to allow the weight to pass through, applying both linear and rotational acceleration during the impact. The acoustic foam and the bottom of the PMMA box also have a 0.6 cm x 1.2 cm opening to allow the passage of the weight drop and avoid re-hits. Please click here to view a larger version of this figure.
This model effectively reproduces many of the biomechanics associated with the injury, including an impact to an unrestrained head, allowing the consequences of both linear forces and rotational acceleration. Using this model in mice, we described the presence of a period of initial transient cutaneous allodynia (CA) lasting several days after mTBI induction revealed by increased frequency of response to tactile stimulation of the periorbital (i.e., cephalic) and hind paw (i.e., extra-cephalic) regions14, suggestive of central sensitization. CA typically persists until day 7 after the mTBI and returns to baseline thresholds by approximately day 14. People with primary headaches often show disruption of central pain modulatory pathways measured by the loss of conditioned pain modulation (CPM)15,16,17,18. Disruption of central pain modulatory pathways has also been observed in individuals with PTH in early and later stages after mTBI19,20,21,22. Correspondingly, we have demonstrated disruption of central pain modulatory pathways following mTBI in this model by the loss of descending noxious inhibitory controls (DNIC), a preclinical measure of CPM in humans13. Loss of DNIC was observed in the first few days after mTBI induction13. Time-dependent allodynia and loss of DNIC following mTBI represent pain-like behaviors in the APTH.
People with migraine and PTH commonly report stress or its relief as a trigger that initiates or exacerbates their attacks21,23,24,26. For this reason, we examined whether stress would elicit allodynia in mice that have recovered from the APTH. Mice were exposed to psychological stress induced by exposure without restraint to bright lights (i.e., bright light stress, BLS) on day 14 following resolution of initial mTBI-induced CA. The BLS reinstated periorbital and hind paw CA only in mice previously subjected to the mTBI14. The model allows adaptation of the ''two-hit'' strategy of hyperalgesic priming27, wherein the initial noxious stimulus induces a vulnerable state by lowering sensory thresholds by the observation of a significant response to a second subthreshold stimulus delivered following resolution of pain from the initial injury27. The occurrence of generalized allodynia elicited by a subthreshold event may therefore represent PPTH-like behaviors.
Our studies using this model of PTH have shown that early peripheral CGRP signaling mediates APTH-like behaviors, the loss of DNIC in mice exposed to mTBI. Additionally, blockade of CGRP signaling early after the mTBI prevented the development of persistent PTH, with a more prominent role during the acute phase of PTH13,14. This model, therefore, may have significant translational relevance and may be valuable for future preclinical studies of pathophysiological mechanisms associated with APTH and PPTH. Additionally, this model may allow the exploration of novel mechanism-based therapeutic approaches. Thus, this protocol aims to describe in detail this unrestrained weight drop model of mTBI.
Protocol
All procedures were approved by the ethical guidelines of the International Association for the Study of Pain regulations on animal welfare and the National Institutes of Health guidelines for the care and use of laboratory animals. The Institutional Animal Care and Use Committee of the University of Arizona previously approved the experimental procedures described in this protocol.
NOTE: The mTBI model described in this protocol is optimized for female or male C57/BL6J12,14,28 and CD-1 IGS (unpublished data) mice between 6-16 weeks old (18-50 g). Implementation of this technique in other strains, weights, or ages will require optimization.
1. Pre mTBI and sham induction preparation
- Preparation for mTBI or sham induction
- Set up the isoflurane inhalational anesthesia station containing an activated charcoal air filter onto a disposable underpad, fix the anesthesia nose cone, get thin pieces of tissue paper (one per animal), with perforation (11 cm x 12 cm) (Figure 2).
- Get a clear tape to fix the tissue paper in the mTBI apparatus (the opening space from the top of the PMMA box). Get a stopwatch to quantify the mTBI/sham induction and righting reflex time (Figure 2).
- Get the mTBI/sham induction-recording sheet of paper (Figure 2). An example of the mTBI/sham induction-recording sheet of paper can be found in Supplementary Material. Bring the mTBI apparatus close to the setup.
- Randomize the animals to receive the mTBI or sham induction.

Figure 2: Preparation for mTBI or sham induction. Materials needed for mTBI and sham induction: An isoflurane inhalational anesthesia station with the nosecone fixed and an activated charcoal air filter, thin pieces of tissue paper, with perforation (11 cm x 12 cm), one per animal, tape to fix the tissue paper in the mTBI apparatus, a stopwatch to quantify the mTBI/sham induction, and the righting time and the mTBI/sham induction-recording sheet of paper. Please click here to view a larger version of this figure.
2. mTBI and sham induction
- mTBI induction
- Get a thin layer of tissue paper and tape it into the opening of the top of the PMMA box of the apparatus (Figure 3A) following steps 2.1.2-2.1.4.
- The top of the PMMA box of the apparatus is not completely fixed. Remove the top, tape the tissue paper, and place it back into the apparatus for induction.
- Tape all four edges of the tissue paper, stretch the paper tightly to support the animal, and ensure minimal resistance to breakthrough after weight drop (Figure 3A).
- Close the opening of the top of the PMMA box with the tissue paper for supporting the animals prior to the impact.
- Place the top of the PMMA box back into the apparatus.
- Turn on the isoflurane inhalational anesthesia station at 5% (i.e., 5 L/min).
- Gently handle the randomly chosen animal that will receive the mTBI and place it into the inhalational anesthesia nose cone.
- Start the stopwatch as soon as the animal stops moving.
- Leave the isoflurane inhalational anesthesia on 5% for 15 s and record it in the recording sheet.
- Switch the volume of isoflurane from 5% to 2% (2 L/min) after the 15 s for an additional 30 s (45 s total) and record it on the recording sheet.
- Remove the animal from the inhalational anesthesia after 45 s and immediately place the animal under the vertical metal guide tube in the apparatus with the ventral surface on the tissue paper in a crucifix position (with both arms open) (Figure 3A).
NOTE: Place the animal's head with the region between the ears and eyes exactly under the vertical metal guide tube (Figure 3B). The weight drop will impact the sagittal suture hitting midway between bregma and lambda. - Test the adequate depth of anesthesia after placing the animal in the proper position (e.g., lack of toe pinch and blink reflex responses). Immediately remove the trigger from the vertical metal guide tube, allowing the weight drop.
NOTE: The weight should drop and hit the animal's head, break the tissue paper allowing the linear and rotational movement of the head and resulting in the animal falling into the acoustic foam cushion (Figure 3C). A representation of the mTBI induction by the weight drop and the linear and rotational movement after the weight drop can be observed in Figure 3. The animal will fall into the acoustic foam cushion right after the weight drop and will stay until the recovery from the anesthesia is observed by moving the hind paw and trying to turn around (i.e., righting reflex time). The animal must be upright to be considered recovered from anesthesia. Do not stop the stopwatch until the animal recovers from the anesthesia. - Record the time the weight drop happened (hit time) on the recording sheet and the righting time (delay time until the animal recovers from the anesthesia).
NOTE: An example of the induction-recording sheet can be observed in Supplementary Material. - Stop the stopwatch and place it back into its home cage after the animal recovers from the anesthesia.
NOTE: It is very important to keep the amount and the duration of the anesthesia to properly control the delay in righting time. mTBI animals should not take more than 5 min to recover from the impact. - Replace the thin layer of tissue paper in between animals.
- Start the procedure with the next animal.
- Clean the equipment with the multi-purpose cleaner solution (3%) after the induction.
- Sham induction
- Follow steps 2.1.1-2.1.11.
NOTE: Again, ensure adequate depth of anesthesia. The animal's head should be placed with the region between the ears exactly under the vertical metal guide tube. There is no weight drop for sham animals (Figure 3D). - Place the sham animal in the proper position onto the tissue paper and immediately take the animal and place it in the supine position (i.e., back down) onto the acoustic foam cushion, similar to how an animal falls after a real mTBI.
NOTE: Righting reflex time is then determined from animal recovery from this position to upright by moving the hind paws and trying to turn around. The animal will not fall into the acoustic foam cushion (Figure 3D). Do not stop the stopwatch until the animal recovers from the anesthesia. - Record the righting time (time that the animal takes to recover from the anesthesia) on the recording sheet of paper.
- Stop the stopwatch and place it back into its home cage after the animal recovers from the anesthesia.
NOTE: Sham animals recover from the anesthesia faster than mTBI animals. Righting time will be shorter than mTBI animals. - Follow steps 2.1.15-2.1.17.
- Follow steps 2.1.1-2.1.11.
- Periorbital and hind paw tactile frequency of response evaluation as APTH and PPTH outcome measurements
NOTE: Periorbital and hind paw frequency of response to tactile stimulation are performed before or after mTBI or sham induction.- Place the mice individually in clear PMMA chambers (10 cm x 10 cm x 20 cm), on top of a wire mesh stand (0.635 cm² grid), for 2 h.
- Apply 0.4 g or 0.6 g mono nylon filaments 10 times to the periorbital (i.e., on the forehead between the eyes) or hind paw region, respectively, for 3 days prior to the baseline collection after the habituation.
- Perform the tactile frequency of response evaluation prior and on days 1, 3, 5, 7, 9, 12, and 14 after the mTBI or sham induction.
- Place the mice individually in the PMMA chambers for 2 h.
- Apply 0.4 g or 0.6 g mono nylon filaments 10 times to the periorbital or hind paw regions, respectively, after the 2 h habituation.
- Apply the filament gently until the filament is slightly arched.
- Score the positive responses after each filament application for the periorbital region (as characterized by facial grooming, head shaking, and/or turning away after filament application) and the hind paw region (characterized by sharp withdrawal of the paw, shaking and/or licking the paw).
NOTE: Increased frequency of response to tactile stimulation represents the development of CA. - Calculate the frequency response by the formula [(number of positive responses x 100)/ 10]. Ensure to include only animals with an average baseline of ~30% frequency of response to complete the experiments13,14.
NOTE: There are different methodologies that might be used for the evaluation of periorbital and hind paw CA in mice, the frequency of response to tactile stimulation is the methodology chosen by this group.
- Environmental bright light stress to restore the CA in mTBI mice
- Keep the mice in their home cages and let them receive one episode of bright light stress (BLS) on day 14 post-mTBI or sham induction as previously described by Navratilova and colleagues14.
NOTE: BLS is induced by two LED lights placed 18 cm from both sides of the home cage to deliver approximately 1,000 lux exposure for 15 min. Food and water should be removed during the BLS exposure. - Place the mice in the individual clear PMMA chambers for CA evaluation as previously described in step 2.3 after the BLS exposure.
- Keep the mice in their home cages and let them receive one episode of bright light stress (BLS) on day 14 post-mTBI or sham induction as previously described by Navratilova and colleagues14.
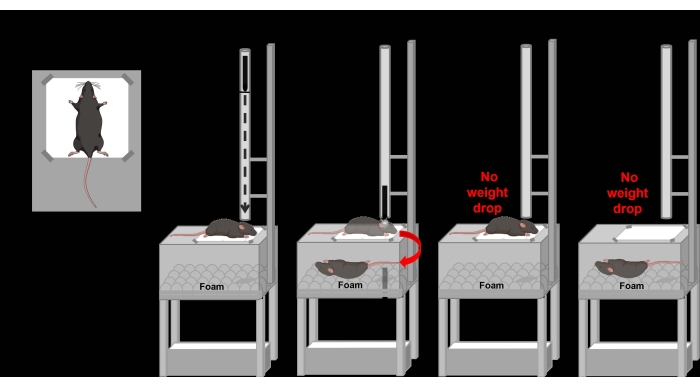
Figure 3: Schematic of mTBI or sham induction. (A) The opening space from the top of the PMMA box with a thin layer of tissue paper stretched and taped to produce a paper stage. The mouse is placed in a crucifix position for mTBI or sham induction. (B) Apparatus for mTBI induction with the animal properly placed onto the paper stage and the head with the ears positioned under the tube guide. The projectile is properly placed to induce the weight drop impact. (C) mTBI induction by pulling the trigger to allow the unrestrained head impact with linear and rotational acceleration with animals falling in cushion foam until complete recovery from the anesthesia. (D) Sham induction by placing the animal onto the paper stage and the head with ears positioned under the tube guide. No projectile is set inside the guide tube, and no head impact is performed. (E) After a sham induction, the animal is placed in the supine position (i.e., back down) onto the acoustic foam cushion until complete recovery from the anesthesia. Please click here to view a larger version of this figure.
Results
Female (Figure 4) and male (Figure 5) mice were submitted to mTBI or sham injury. Development of immediate and transient periorbital (cephalic) (Figure 4A and Figure 5A) and hind paw (extra-cephalic) (Figure 4C and Figure 5C) CA was observed only in mice submitted to mTBI, demonstrated by increased frequency of response after tactile stimulatio...
Discussion
Herein, we describe a mouse model of mTBI in which the head is unfixed, reproducing many biomechanical aspects of common human mild head injuries, including closed-skull, blunt force direct impact, and linear, rotational, acceleration, and shearing forces from unrestrained head movement. It is suggested that these similarities of the model to human concussion might allow for increased relevance of investigations of pathophysiological mechanisms associated with APTH and the transition to PPTH. The injury produced by this ...
Disclosures
Caroline Machado Kopruszinski, Janice Oyarzo, and Edita Navratilova declare that they have no personal, financial, or relational conflicts of interest with this work. Trent Anderson has received compensation from Novartis for PTH consultancy last year and has a material agreement with Amgen. Frank Porreca has served as a consultant or received research funding from Voyager, SiteOne Therapeutics, Nektar, Amgen, Acadia, Blackthorn, Teva, Eli Lilly, Hoba, Allergan, Ipsen, and Proximagen and is a founder of Catalina Pharma.
Acknowledgements
We kindly thank Luiz Henrique Moreira de Souza for the supporting illustrations.
Materials
| Name | Company | Catalog Number | Comments |
| Clear PMMA chambers (10 x 10 x 20 cm) | NA | NA | in house |
| Disposable underpad | VWR | 56617-014 | Can be replaced by any brand of laboratory disposable underpad |
| Fluriso Isoflurane | Vet One | 502017 | Can be replaced by any animal laboratory isuflurane inhalational anesthesia |
| Inhalational anesthesia station | Parkland Scientific | PM1002 | Can be replaced by any animal laboratory inhalational anesthesia station |
| LED lights for BLS exposure | |||
| Mesh stand (0.635 cm² grid) for behavioral testing | NA | NA | in house |
| Mono nylon filament for hindpaw region (0.6 g/3.84) | North Coast medical | NC12775-07 | |
| Mono nylon filament for periorbital region (0.4 g/3.61) | North Coast medical | NC12775-06 | |
| mTBI/Sham apparatus | NA | NA | in house |
| mTBI/sham induction-recording sheet of paper | NA | NA | Prepare the sheet according to the Supplementary Material |
| Pen | Fisher Scientific | NC1836221 | Any brand of pen |
| Scotch Magic Clear tape 3M | Fisher Scientific | 50-205-2453 | Can be replaced by any brand of clear tape |
| Stopwatch | Fisher Scientific | 06-662-56 | Can be replaced by any brand of stopwatch |
| Tissue paper | Kleenex/Fisher Scientific | 15-235-130 | Can be replaced by any brand tissue paper (4.4” x 8.4”) |
References
- Ashina, H., et al. Post-traumatic headache: epidemiology and pathophysiological insights. Nature Reviews Neuroscience. 15 (10), 607-617 (2019).
- Larsen, E. L., et al. Acute and preventive pharmacological treatment of post-traumatic headache: a systematic review. Journal of Headache Pain. 20 (1), 98 (2019).
- Schwedt, T. J. Post-traumatic headache due to mild traumatic brain injury: Current knowledge and future directions. Cephalalgia. 41 (4), 464-471 (2021).
- Dewan, M. C., et al. Estimating the global incidence of traumatic brain injury. Journal of Neurosurgery. , 1-18 (2018).
- Skandsen, T., et al. Incidence of mild traumatic brain injury: A prospective hospital, emergency room and general practitioner-based study. Frontiers of Neurology. 10, 638 (2019).
- Lucas, S., Hoffman, J. M., Bell, K. R., Dikmen, S. A prospective study of prevalence and characterization of headache following mild traumatic brain injury. Cephalalgia. 34 (2), 93-102 (2014).
- Bree, D., Levy, D. Strides toward better understanding of post-traumatic headache pathophysiology using animal models. Current Pain and Headache Reports. 22 (10), 67 (2018).
- Schwedt, T. J. Structural and functional brain alterations in post-traumatic headache attributed to mild traumatic brain injury: A narrative review. Frontiers in Neurology. 10, 615 (2019).
- Ashina, H., et al. Post-traumatic headache attributed to traumatic brain injury: classification, clinical characteristics, and treatment. Lancet Neurology. 20 (6), 460-469 (2021).
- Bodnar, C. N., Roberts, K. N., Higgins, E. K., Bachstetter, A. D. A systematic review of closed head injury models of mild traumatic brain injury in mice and rats. Journal of Neurotrauma. 36 (11), 1683-1706 (2019).
- Goddeyne, C., Nichols, J., Wu, C., Anderson, T. Repetitive mild traumatic brain injury induces ventriculomegaly and cortical thinning in juvenile rats. Journal of Neurophysiology. 113 (9), 3268-3280 (2015).
- Kane, M. J., et al. A mouse model of human repetitive mild traumatic brain injury. Journal of Neuroscience Methods. 203 (1), 41-49 (2012).
- Kopruszinski, C. M., et al. CGRP monoclonal antibody prevents the loss of diffuse noxious inhibitory controls (DNIC) in a mouse model of post-traumatic headache. Cephalalgia. 41 (6), 749-759 (2021).
- Navratilova, E., et al. CGRP-dependent and independent mechanisms of acute and persistent post-traumatic headache following mild traumatic brain injury in mice. Cephalalgia. 39 (14), 1762-1775 (2019).
- Lockwood, S., Dickenson, A. H. What goes up must come down: insights from studies on descending controls acting on spinal pain processing. Journal of Neural Transmission. 127 (4), 541-549 (2020).
- Nir, R. R., Yarnitsky, D. Conditioned pain modulation. Current Opinion in Supportive and Palliative Care. 9 (2), 131-137 (2015).
- Williams, A. E., et al. Impairment of inhibition of trigeminal nociception via conditioned pain modulation in persons with migraine headaches. Pain Medicine. 20 (8), 1600-1610 (2019).
- Yarnitsky, D. Conditioned pain modulation (the diffuse noxious inhibitory control-like effect): its relevance for acute and chronic pain states. Current Opinion in Anaesthesiology. 23 (5), 611-615 (2010).
- Carey, C., Saxe, J., White, F. A., Naugle, K. M. An exploratory study of endogenous pain modulatory function in patients following mild traumatic brain injury. Pain Medicine. 20 (11), 2198-2207 (2019).
- Defrin, R. Chronic post-traumatic headache: clinical findings and possible mechanisms. The Journal of Manual & Manipulative Therapy. 22 (1), 36-44 (2014).
- Defrin, R., Riabinin, M., Feingold, Y., Schreiber, S., Pick, C. G. Deficient pain modulatory systems in patients with mild traumatic brain and chronic post-traumatic headache: implications for its mechanism. Journal of Neurotrauma. 32 (1), 28-37 (2015).
- Irvine, K. A., Clark, J. D. Chronic pain after traumatic brain injury: Pathophysiology and pain mechanisms. Pain Medicine. 19 (7), 1315-1333 (2018).
- Borsook, D., Maleki, N., Becerra, L., McEwen, B. Understanding migraine through the lens of maladaptive stress responses: a model disease of allostatic load. Neuron. 73 (2), 219-234 (2012).
- Lipton, R. B., et al. Reduction in perceived stress as a migraine trigger: testing the "let-down headache" hypothesis. Neurology. 82 (16), 1395-1401 (2014).
- Robbins, L. Precipitating factors in migraine: a retrospective review of 494 patients. Headache. 34 (4), 214-216 (1994).
- Silverberg, N. D., Martin, P., Panenka, W. J. Headache trigger sensitivity and avoidance after mild traumatic brain injury. Journal of Neurotrauma. 36 (10), 1544-1550 (2019).
- Kandasamy, R., Price, T. J. The pharmacology of nociceptor priming. Handbook of Experimental Pharmacology. 227, 15-37 (2015).
- Khuman, J., et al. Low-level laser light therapy improves cognitive deficits and inhibits microglial activation after controlled cortical impact in mice. Journal of Neurotrauma. 29 (2), 408-417 (2012).
- Bree, D., Levy, D. Development of CGRP-dependent pain and headache related behaviours in a rat model of concussion: Implications for mechanisms of post-traumatic headache. Cephalalgia. 38 (2), 246-258 (2018).
- Kopruszinski, C. M., et al. Prevention of stress- or nitric oxide donor-induced medication overuse headache by a calcitonin gene-related peptide antibody in rodents. Cephalalgia. 37 (6), 560-570 (2017).
- Moye, L. S., et al. The development of a mouse model of mTBI-induced post-traumatic migraine, and identification of the delta opioid receptor as a novel therapeutic target. Cephalalgia. 39 (1), 77-90 (2019).
- Mychasiuk, R., et al. A novel model of mild traumatic brain injury for juvenile rats. Journal of Visualized Experiments: JoVE. (94), e51820 (2014).
- Meaney, D. F., Smith, D. H. Biomechanics of concussion. Clinical Journal of Sports Medicine. 30 (1), 19-31 (2011).
- Stemper, B. D., et al. Prediction of post-concussive behavioral changes in a rodent model based on head rotational acceleration characteristics. Annals of Biomedical Engeneering. 44 (11), 3252-3265 (2016).
- Stemper, B. D., et al. Head rotational acceleration characteristics influence behavioral and diffusion tensor imaging outcomes following concussion. Annals of Biomedical Engeneering. 43 (5), 1071-1088 (2015).
- Bharadwaj, V. N., et al. Blood-brainbarrier disruption dictates nanoparticle accumulation following experimental brain injury. Nanomedicine. 14 (7), 2155-2166 (2018).
- Price, T. J., Inyang, K. E. Commonalities between pain and memory mechanisms and their meaning for understanding chronic pain. Progress in Molecular Biology and Translational Science. 131, 409-434 (2015).
- Reichling, D. B., Levine, J. D. Critical role of nociceptor plasticity in chronic pain. Trends in Neuroscience. 32 (12), 611-618 (2009).
- Ashkenazi, A., Silberstein, S., Jakubowski, M., Burstein, R. Improved identification of allodynic migraine patients using a questionnaire. Cephalalgia. 27 (4), 325-329 (2007).
- LoPinto, C., Young, W. B., Ashkenazi, A. Comparison of dynamic (brush) and static (pressure) mechanical allodynia in migraine. Cephalalgia. 26 (7), 852-856 (2006).
- Yilmaz, T., et al. Risk factors and outcomes associated with post-traumatic headache after mild traumatic brain injury. Emergency Medicine Journal. 34 (12), 800-805 (2017).
- Bartley, E. J., Fillingim, R. B. Sex differences in pain: a brief review of clinical and experimental findings. British Journal of Anaesthesia. 111 (1), 52-58 (2013).
- Herson, P. S., Koerner, I. P., Hurn, P. D. Sex, sex steroids, and brain injury. Seminars in Reproductive Medicine. 27 (3), 229-239 (2009).
- Arain, M., et al. Maturation of the adolescent brain. Neuropsychiatric Disease and Treatment. 9, 449-461 (2013).
- Castelli, V., et al. Neuronal cells rearrangement during aging and neurodegenerative disease: metabolism, oxidative stress and organelles dynamic. Frontiers in Molecular Neuroscience. 12, 132 (2019).
- Peters, R. Ageing and the brain. Postgraduate Medical Journal. 82 (964), 84-88 (2006).
- Gonzalez-Roldan, A. M., et al. Age-related changes in pain perception are associated with altered functional connectivity during resting state. Frontiers in Aging Neuroscience. 12, 116 (2020).
- Loizzo, A., Loizzo, S., Capasso, A. Neurobiology of pain in children: an overview. TheOpen Biochemistry Journal. 3, 18-25 (2009).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved