A subscription to JoVE is required to view this content. Sign in or start your free trial.
DNA Extraction and Comparison Between Old and Fresh Necrophilic Fly Samples
* These authors contributed equally
In This Article
Summary
This article describes a protocol for fresh and old necrophilic fly DNA extraction using a modified common DNA extraction kit.
Abstract
A total of five samples of Chrysomya megacephala samples - three fresh samples, one sample stored in alcohol for 2 years, and one sample stored in dry sealed storage for 2 years protected from light only - were selected to investigate whether a blood DNA extraction kit could extract DNA from necrophilous flies and to determine whether alcohol could prolong the preservation of necrophilous flies' DNA. First, the blood DNA extraction kit was used to extract DNA from their thorax tissues. Then, the DNA purity and concentration were examined using a microplate reader and a fluorometer. Finally, PCR amplification and electrophoresis of the extracted DNA were done with necrophilic fly-specific primers located in the mitochondrial CO I gene sequence. The results showed that the DNA purity of all samples was greater than 2.0. The DNA concentration was observed to be of the following order: fresh samples > alcohol-preserved old samples > untreated, old samples. All samples had specific electrophoretic bands after PCR amplification. In conclusion, a blood DNA extraction kit can be used to extract DNA from necrophilic flies successfully, and the DNA concentration of fresh fly samples is greater than that of old fly samples. The flies can be stored in alcohol for a long time.
Introduction
The inference of the time of death has always been one of the key and difficult issues to be resolved in judicial practice. In the practice of forensic science, for a criminal case, determining postmortem interval plays a crucial role in deducing the time of the crime, locking in the suspect, and narrowing the scope of the investigation. Traditional methods of inferring the time of death are based primarily on early postmortem phenomena, which can generally only be used to infer the time of death within 24 h. However, the long time of death cannot be determined by postmortem phenomena. It is now generally recognized in the forensic science community that forensic entomology has the potential to be an effective method of inferring a longer time of death.
Necrophagous insects are named for their larvae that feed on corpses. Among them, whenever a corpse is present, the adult necrophilous flies will be the first to reach the corpse and lay their eggs or larvae. The larvae feed on corpse tissue and mature into pupae, which fledge into adults. Necrophilic flies play a huge role in the practice of forensic science because of their regular life cycle and geographic distribution, for example, deducting the time of death and determining the place of death1,2. However, the barrier to the use of necrophilous flies in forensic practice is species identification. Morphological methods are still used as the authoritative method for species identification of necrophilous flies. However, morphological species identification requires a high degree of insect integrity. If the flies were in different developmental stages, or due to morphological changes caused by environmental choices, those all make morphological examination more difficult. In particular, the morphology of pupae and pupal shells, which are most often found at the crime scene, is barely recognizable. This, together with the scarcity of morphological experts, makes huge difficulty to the identification of species morphologically. Therefore, the application of molecular biology methods for species identification of flies has emerged, which reduces the difficulty of morphological identification and is independent of developmental stage3,4,5,6,7.
The first step in species identification of necrophilous flies based on molecular biology methods is the efficient extraction of DNA, while specialized kits for insect DNA extraction are not commonly available currently. And how to use common blood kits for DNA extraction of necrophilic flies becomes more forensically relevant. Necrophilic flies found at crime scenes are often mutilated or have undergone decay and DNA degradation. Fly samples are not immediately available for DNA extraction after acquisition, or if possible, complete samples need to be retained due to evidence preservation requirements. Based on the above requirements, in this study, we applied the blood DNA kit based on proteinase K digestion, and selected three fresh Chrysomya megacephala (Diptera, Lepidoptera) samples (Figure 1), one old C. megacephala sample placed in alcohol for two years, and one C. megacephala sample placed in light-protected storage for two years, weighed each of the five samples, elected the insect thorax tissue for DNA extraction. Comparing the quality and purity of DNA extracted from the old and new samples, PCR amplification and electrophoresis of the extracted DNA were performed with necrophilic fly-specific primers located in the mitochondrial CO 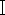 gene sequence.
gene sequence.
Protocol
NOTE: A total of five samples of C. megacephala samples were used in this protocol-three fresh samples (Fresh 1, Fresh 2, and Fresh 3), one sample stored in alcohol for 2 years (Old 1), and one sample stored in dry sealed storage for 2 years protected from light only (Old 2). Samples must be labeled according to the experimental requirements.
1. General sample storage and preparations
- Sample selection and preparation
NOTE: Be sure to work in the fume hood when applying ethyl acetate to execute necrophagous flies. Remove the liquid from the surface of the fly before weighing.- Fresh samples: Place three collected fly samples in gas vials after killing them with ethyl acetate. Weigh and name the samples as Fresh 1, Fresh 2, and Fresh 3 and use them immediately in experiments.
NOTE: The fresh fly samples in this protocol had masses of 56.8 mg, 49.3 mg, and 52.1 mg. - Old sample 1: Take out fly samples killed by ethyl acetate and stored in alcohol-filled centrifuge tubes for 2 years in airtight containment, protected from light at room temperature. Weigh the sample after adsorbing the surface liquid with filter paper; call it Old 1.
NOTE: The mass of Old 1 was 42.3 mg in this protocol. - Old sample 2: Take out the fly sample killed by ethyl acetate and stored in a centrifuge tube for 2 years in airtight containment, protected from light at room temperature. Weigh and call it Old 2.
- Fresh samples: Place three collected fly samples in gas vials after killing them with ethyl acetate. Weigh and name the samples as Fresh 1, Fresh 2, and Fresh 3 and use them immediately in experiments.
- Preparation before extraction
- Add a small amount of liquid nitrogen to cool a stainless steel container; wait until all liquid nitrogen has evaporated.
- Put a fly sample in the container; pour in the liquid nitrogen slowly to prevent the liquid nitrogen from splashing and impacting the insect sample.
- Freeze the samples in liquid nitrogen. Remove samples after the liquid nitrogen evaporates.
- Separate the thorax individually, place in a 2 mL centrifuge tube, and cut into the smallest possible slices.
NOTE: Cut the sample into small pieces so that it can be fully cleaved, and DNA can be released in large quantities. - Repeat the above steps for each fly, individually freeze, and then remove to a new centrifuge tube to prevent DNA cross-contamination.
2. DNA extraction
NOTE: All centrifugation steps must be carried out at room temperature (15-25 °C) in a microcentrifuge. All steps must be performed in strict adherence to the principles of asepsis and to avoid cross-contamination.
- Tissue lysis
- Add 180 µL of tissue lysis buffer to a 2 mL centrifuge tube containing a clipped sample.
- Add 20 µL of proteinase K. Mix thoroughly by vortexing at 3 000 rpm for 10 s and incubate at 56 °C until the insect tissues are completely lysed.
NOTE: Add more Proteinase K should be if the tissue is not fully digested. Lysis is usually complete in 1-3h, also can extend lysis time overnight.
- DNA precipitation
- Vortex at 3,000 rpm for 15 s. Add 200 µL lysis buffer to the sample, and mix thoroughly by vortexing at 3,000 rpm for 15 s.
- Add 200 µL of ethanol (96-100%) and mix again thoroughly by vortexing at 3,000 rpm for 15 s.
NOTE: Lysis buffer and ethanol can be preadded together in one step to save time. A white precipitate may form on the addition of lysis buffer and ethanol.
- Pipet the mixture into the DNA adsorption column placed in a 2 mL collection tube. Centrifuge at 6,000 × g for 1 min. Discard flowthrough and collection tube.
- Place the DNA adsorption column from the previous step in a new 2 mL collection tube, add 500 µL of protein removal buffer, and centrifuge at 6,000 × g for 1 min. Discard flowthrough and collection tube.
- Transfer the DNA adsorption column to a new 2 mL collection tube, add 500 µL of desalination buffer, and centrifuge at 20,000 × g for 3 min to dry the DNA adsorption column membrane. Discard flowthrough and collection tube.
NOTE: This centrifugation step ensures the removal of any residual ethanol before the following elution. If there is any carryover of ethanol, centrifuge again at 20,000 × g for 1 min. - Place the DNA adsorption column in a clean 2 mL microcentrifuge tube, directly pipet 100 µL of DNA elution buffer onto the column membrane, incubate at room temperature for 1 min, and then centrifuge at 6,000 × g for 1 min to elute.
NOTE: Elution with 100 µL (instead of 200 µL) increases the final DNA concentration in the eluate but also decreases the overall DNA yield. To maximize the DNA yield, repeat elution once as described in step 2.8. - Store the DNA samples at 4 °C (up to 1 week) or -80 °C for long-term storage.
3. DNA concentration detection
- Determine the values of OD 260 and OD 280 to compare DNA purity and nucleic acid concentration by microplate reader. Repeat three times on each sample.
- Determine the concentration of the extracted DNA by fluorometer using its companion kit, with three replicates for each sample.
- Take 190 µL of working solution into two 0.5 mL centrifuge tubes, take 10 µL each of Standard #1 and Standard #2, add them to the working solutions, and keep them away from light for not less than 15 min.
NOTE: Standard #1 and Standard #2 are accurately calibrated for a certain concentration of DNA. Standard solutions should be accurately configured to ensure the formation of an accurate standard curve. Standard #1 is a 0.2 ng/µL double-stranded DNA (dsDNA) sample; standard #2 is 2,000 ng/µL. The working solution is a dye that binds specifically to dsDNA. - Take 2 µL of test DNA and mix it with 198 µL of working solution in a 0.5 mL centrifuge tube, and keep away from light for not less than 15 min.
NOTE: If the DNA concentration is low, add 5 µL of test DNA with 195 µL of working solution. - After 15 min, turn on the instrument and select dsDNA Concentration Measurement | Long Range. First, test Standard #1, and then, Standard #2 to obtain the standard curve.
NOTE: The standard solution must be used on the same day after it is configured; leaving it for too long will result in a large error in the standard curve. - Place the samples to be tested in the assay wells one by one, adjust the DNA spiking volume to 2 µL, repeat the measurement three times, take the average value, and record.
- Take 190 µL of working solution into two 0.5 mL centrifuge tubes, take 10 µL each of Standard #1 and Standard #2, add them to the working solutions, and keep them away from light for not less than 15 min.
4. PCR and electrophoretic detection
NOTE: The mitochondrial CO 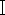 gene sequence with a length of 278 bp was taken for PCR amplification with forward primer C1-J-2495: CAGCTACTTTATGAGCTTTAGG, and reverse primer C1-N-2800: CATTTCAAGCTGTGTAAGCATC8, and then 2% agarose gel electrophoresis done to verify the amplification effect and amplification products' length was verified.
gene sequence with a length of 278 bp was taken for PCR amplification with forward primer C1-J-2495: CAGCTACTTTATGAGCTTTAGG, and reverse primer C1-N-2800: CATTTCAAGCTGTGTAAGCATC8, and then 2% agarose gel electrophoresis done to verify the amplification effect and amplification products' length was verified.
- Set up 20 µL of the PCR amplification system by adding 10 µL of 2x Taq mix, 1 µL of each primer, 5 µL of ddH2O, and 3 µL of template DNA; mix well for 10 s in a centrifuge.
- Use the following PCR cycling parameters: predenaturation 95 °C for 10 min; denaturation 95 °C for 30 s, annealing 48 °C for 30 s, extension 72 °C for 45 s for a total of 40 cycles; and final extension 72 °C for 10 min.
- Subject the products after PCR to 2% agarose gel electrophoresis, and after electrophoresis, place them on a gel imaging system for photo analysis.
Results
We used a microplate reader to measure the values of OD260 and OD280 of the extracted DNA solution and then obtained the OD260/OD280 values to evaluate the purity of the DNA. The OD260/OD280 of all fresh samples and old sample 1 was greater than 2. The OD260/OD280 of old sample 2 had a maximum value of 2.187 although the average was 1.753, and its three measurements fluctuated greatly due to the small (≤0.01) values of both its OD280 and OD280 (Table 1). Based on the value of OD260 obtained, we est...
Discussion
DNA extraction is the most critical link in the molecular identification of necrophilous flies, and its extraction method and the quality of the extracted DNA directly affect subsequent detection. In this experiment, we successfully extracted DNA from necrophilous flies by using a common blood kit, without the need to purchase a special insect DNA extraction kit, which makes insect DNA extraction easier to accomplish.
The surface of flies is rich in chitin, especially in the larval and pupal s...
Disclosures
The authors declare that they have no competing financial interests.
Acknowledgements
This work is supported by the National Natural Science Foundation of China (82060341,81560304) and by the Academician Innovation Platform Scientific Research Project of Hainan Province (YSPTZX202134).
Materials
| Name | Company | Catalog Number | Comments |
| Buffer AE | Qiagen | 172026832 | DNA elution buffer |
| Buffer AL | Qiagen | 172028374 | lysis buffer |
| Buffer ATL | Qiagen | 172028162 | tissue lysis buffer |
| Buffer AW1 | Qiagen | 172028760 | protein removal buffer |
| Buffer AW2 | Qiagen | 57203108 | desalination buffer |
| C1-J-2495 | Taihe Biotechnology | TW21109216 | forword primer |
| C1-N-2800 | Taihe Biotechnology | TW21109217 | reverse primer |
| D2000 DNA ladder | Real-Times(Beijing) Biotechnology | RTM415 | Measure the position of electrophoretic bands |
| DNeasy Mini spin column | Qiagen | 166050343 | DNA adsorption column |
| Dry Bath Incubator | Miulab | DKT200-2D | used for heating |
| MIX-30S Mini Mixer | Miulab | MUC881206 | oscillatory action |
| Proteinase K | Qiagen | 172026218 | Inactivation of intracellular nucleases and other proteins |
| Qubit 3.0 Fluorometer | Thermo Fisher Scientific | 2321611188 | |
| Speed Micro-Centrifuge | Scilogex | 9013001121 | centrifuge |
| Standard#1 | Thermo Fisher Scientific | 2342797 | |
| Standard#2 | Thermo Fisher Scientific | 2342797 | |
| Tanon 3500R Gel Imager | Tanon | 16T5553R-455 | gel imaging |
| Taq Mix Pro | Monad | 00007808-140534 | PCR Mix |
| Taq Mix Pro | Monad | 00007808-140534 | PCR Mix |
| Thermo Cycler | Zhuhai Hema | VRB020A | ordinary PCR |
| Working Solution | Thermo Fisher Scientific | 2342797 |
References
- Amendt, J., Richards, C. S., Campobasso, C. P., Zehner, R., Hall, M. J. Forensic entomology: applications and limitations. Forensic Sci Med Pathol. 7 (4), 379-392 (2011).
- Wydra, J., Matuszewski, S. The optimal post-eclosion interval while estimating the post-mortem interval based on an empty puparium. Forensic Sci Med Pathol. 17 (2), 192-198 (2021).
- Mazzanti, M., Alessandrini, F., Tagliabracci, A., Wells, J. D., Campobasso, C. P. DNA degradation and genetic analysis of empty puparia: genetic identification limits in forensic entomology. Forensic Sci Int. 195 (1-3), 99-102 (2010).
- Odeniran, P. O., et al. molecular identification and distribution of Trypanosome-transmitting Dipterans from cattle settlements in Southwest Nigeria. Acta Parasitol. 66 (1), 116-128 (2021).
- Wells, J. D., Stevens, J. R. Application of DNA-based methods in forensic entomology. Annu Rev Entomol. 53, 103-120 (2008).
- Kotrba, M., et al. DNA barcoding in forensic entomology-establishing a DNA reference library of potentially forensic relevant arthropod species. J Forensic Sci. 64 (2), 593-601 (2019).
- Weedon, M. N., et al. Use of SNP chips to detect rare pathogenic variants: Retrospective, population based diagnostic evaluation. BMJ. 372, 214 (2021).
- Cai, J. -. F., et al. The availability of mitochondrial DNA cytochrome oxidasegene for the distinction of forensically important flies in China. Acta Entomologica Sinica. 48 (3), 380-385 (2005).
- Khayrova, A., Lopatin, S., Varlamov, V. Obtaining chitin, chitosan and their melanin complexes from insects. Int J Biol Macromol. 167, 1319-1328 (2021).
- Guo, Y. Q., et al. Comparison of three DNA extracting methods from different parts of common necrophagous flies. J Henan Univ Sci Tech (Med Sci). 36 (02), 130-133 (2018).
- Du, H., Xie, H., Ma, M., Igarashi, Y., Luo, F. Modified methods obtain high-quality DNA and RNA from anaerobic activated sludge at a wide range of temperatures. J Microbiol Methods. 199, 106532 (2022).
- Sukumaran, S. Concentration determination of nucleic acids and proteins using the micro-volume BioSpec-nano-spectrophotometer. J Vis Exp. (48), e2699 (2011).
- Masago, K., et al. Comparison between fluorimetry (Qubit) and spectrophotometry (NanoDrop) in the quantification of DNA and RNA extracted from frozen and FFPE tissues from lung cancer patients: A real-world use of genomic tests. Medicina (Kaunas). 57 (12), 1375 (2021).
- Ponti, G., et al. The value of fluorimetry (Qubit) and spectrophotometry (NanoDrop) in the quantification of cell-free DNA (cfDNA) in malignant melanoma and prostate cancer patients. Clin Chim Acta. 479, 14-19 (2018).
- Adams, Z. J., Hall, M. J. Methods used for the killing and preservation of blowfly larvae, and their effect on post-mortem larval length. Forensic Sci Int. 138 (1-3), 50-61 (2003).
- Fliegerova, K., et al. Effect of DNA extraction and sample preservation method on rumen bacterial population. Anaerobe. 29, 80-84 (2014).
- Rodrigues, B. L., Galati, E. A. B. Molecular taxonomy of phlebotomine sand flies (Diptera, Psychodidae) with emphasis on DNA barcoding: A review. Acta Trop. 238, 106778 (2023).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved