A subscription to JoVE is required to view this content. Sign in or start your free trial.
Volatile Sex Pheromone Extraction and Chemoattraction Assay in Caenorhabditis elegans
In This Article
Summary
This protocol establishes methods for extracting and quantifying responses to the volatile sex pheromone in C. elegans, providing tools to study chemical communication and navigation trajectory.
Abstract
Chemical communication is vital in organismal health, reproduction, and overall well-being. Understanding the molecular pathways, neural processes, and computations governing these signals remains an active area of research. The nematode Caenorhabditis elegans provides a powerful model for studying these processes as it produces a volatile sex pheromone. This pheromone is synthesized by virgin females or sperm-depleted hermaphrodites and serves as an attractant for males.
This protocol describes a detailed method for isolating the volatile sex pheromone from several C. elegans strains (WT strain N2, daf-22, and fog-2) and C. remanei. We also provide a protocol for quantifying the male chemotaxis response to the volatile sex pheromone. Our analysis utilizes measurements such as chemotaxis index (C.I.), arrival time (A.T.), and a trajectory plot to compare male responses under various conditions accurately. This method allows for robust comparisons between males of different genetic backgrounds or developmental stages. Furthermore, the experimental setup outlined here is adaptable to investigating other chemoattraction chemicals.
Introduction
The interplay between chemical communication and reproductive success is a fundamental principle across the animal kingdom1,2,3,4,5,6,7,8,9,10. Sex pheromones trigger a wide array of sexually dimorphic behaviors essential for locating mates, coordinating the steps involved in finding and attracting a partner, and ultimately promoting the propagation of a species11,12,13,14,15,16,17. Significant progress has been made in understanding pheromone signaling, but the molecular mechanisms, neural circuits, and computational processes governing these interactions often remain incompletely defined18,19,20,21,22,23,24,25,26.
The nematode Caenorhabditis elegans provides a powerful model for dissecting these questions. Notably, C. elegans exhibits an unusual reproductive strategy-hermaphrodites can self-fertilize but also outcross with males27,28,29,30,31,32,33. This flexibility requires a robust communication system to signal reproductive status. C. elegans is known for its well-characterized water-soluble pheromones, the ascarosides, which play varied roles in development, behavior, and social interactions. Recent discoveries have unveiled a distinct class of volatile sex pheromones employed by nematodes. These pheromones are specifically produced by sexually mature C. elegans and C. remanei virgin females and sperm-depleted hermaphrodites, serving as an attractant for adult males29,34,35. This attractant exhibits remarkable sexual dimorphism in its production and perception. The female somatic gonad governs the synthesis of this volatile sex pheromone, and production dynamically reflects reproductive status, ceasing upon mating and resuming several hours later29,34.
Understanding nematode sex pheromone communication provides insights into the evolution of chemical communication systems, the interplay between reproductive state and behavior, and the mechanisms underlying sexually dimorphic neural processing24,26,36,37,38,39. Studies implicate the amphid neuron AWA in males as critical for pheromone detection, with the G-protein-coupled receptor SRD-1 playing a key role in pheromone detection in males24. C. elegans is well-suited for studying animal chemical communication, especially sex pheromone signaling, due to its reliance on the olfactory system for mate-searching. While much is known about ascaroside signaling, the volatile sex pheromone system offers unique opportunities for comparison25,26,36,40,41,42,43,44,45,46,47,48,49,50,
51,52,53,54,55,56,57. Moreover, C. elegans is a powerful genetic model organism due to its fully sequenced genome, clearly defined cellular lineage, and well-characterized olfactory mutants.
However, the complete neural circuitry involved in processing this pheromone, the computations that translate its perception into targeted mate-searching behaviors, and its biosynthesis regulation remain to be fully elucidated. Further investigations into these processes are crucial for understanding the diverse mechanisms governing animal communication and reproductive behaviors. The identification of key genes involved in pheromone synthesis, secretion, and perception promises to unveil novel molecular players in animal communication. The assays described here provide a basis to address these questions.
Protocol
1. Crude sex pheromone extraction from females and hermaphrodites
- Protocol for C. elegans Synchronization
- Preparation of adult females or hermaphrodites
- Monitor culture plates daily until a large population of adult female/hermaphrodites exists and the OP50 food source depletes. Using fog-2 C. elegans and WT C. remanei females for crude sex pheromone extraction, prepare synchronized eggs from the mated females.
NOTE: In this protocol, C. elegans fog-2 mutant XX animals, which do not produce self-sperm, are referred to as C. elegans females.
- Monitor culture plates daily until a large population of adult female/hermaphrodites exists and the OP50 food source depletes. Using fog-2 C. elegans and WT C. remanei females for crude sex pheromone extraction, prepare synchronized eggs from the mated females.
- Washing and pelleting worms
- Wash adult worms off a mixed population plate with M9 buffer. Collect worm suspension into a 1.5 mL microcentrifuge tube. Centrifuge at 1,500 × g for 0.5-1 min to pellet the worms.
NOTE: Precleaning for heavy contamination (optional): Let the worms settle in the tube if heavily contaminated. Pipette out the supernatant to remove bacteria and repeat until the supernatant becomes clear.
- Wash adult worms off a mixed population plate with M9 buffer. Collect worm suspension into a 1.5 mL microcentrifuge tube. Centrifuge at 1,500 × g for 0.5-1 min to pellet the worms.
- Bleaching
- Add 200 µL of M9 buffer to the worm pellet. Prepare lysis buffer by mixing household bleach and 1 M NaOH in a 1:1 ratio. Add 500 µL of this lysis buffer to the mixture, vortex for 10 s, and then pause to monitor the lysis condition under a dissecting microscope. Repeat this process of 10 s vortexes followed by observation until the adult worms are fully lysed.
- Stopping bleaching and pelleting the embryos
- When adult worms are lysed into small fragments (but not completely dissolved), immediately add 500 µL of M9 buffer to slow the reaction. Centrifuge at 15,000 × g for 30-60 s to pellet the embryos.
NOTE: Over-lysis can damage the embryos. Stop the reaction when the adult worm bodies have broken down into small fragments. Lysis continues during the centrifuge and washing steps until all the lysis buffer is removed.
- When adult worms are lysed into small fragments (but not completely dissolved), immediately add 500 µL of M9 buffer to slow the reaction. Centrifuge at 15,000 × g for 30-60 s to pellet the embryos.
- Wash the embryos 5x with 1 mL of M9 buffer, centrifuging at 15,000 × g for 30-60 s after each wash. Remove the supernatant after centrifugation.
- Synchronization
- Resuspend the embryos in 800 µL of M9 buffer in a 1.5 mL microcentrifuge tube. Rotate the tube at 20 °C for 12-15 h for L1 hatching and worm arrest at the L1 stage due to no food supply. Release and culture the worms at 20° C for 3 days until they reach the L4 stage.
- Preparation of adult females or hermaphrodites
2. Crude sex pheromone extraction from one-day-old virgin females (Figure 1A)
NOTE: We adopt a previously established protocol24 to extract sex pheromones from one-day-old virgin fog-2 (feminization of germline) mutant females of C. elegans and WT females of C. remanei.
- Preparation of adult females
- Select and isolate approximately 200 L4 stage fog-2 females 1 day prior to pheromone extraction. To ensure only virgin females are collected, separate these L4 fog-2 females carefully by sex and distribute them onto three separate 6 cm OP50 NGM plates. Place a small amount of OP50 bacteria in the center of each plate.
NOTE: Distribute 200 L4 female worms across three separate plates to avoid the risk of accidentally introducing a male worm to the plate. If a male is picked, it is likely to mate with many females, resulting in most of the females being fertilized by the following day when they become adults. Fertilized females do not emit volatile sex pheromones until sperm is exhausted, which affects the extraction outcomes. The limited food source restricts the hermaphrodites to a smaller area in the center of the plate, minimizing the chance of escape from the plate during the isolation period.
- Select and isolate approximately 200 L4 stage fog-2 females 1 day prior to pheromone extraction. To ensure only virgin females are collected, separate these L4 fog-2 females carefully by sex and distribute them onto three separate 6 cm OP50 NGM plates. Place a small amount of OP50 bacteria in the center of each plate.
- Extraction process
- On the extraction day (day 1 of reproductive maturity, 3 days after worms were released from L1 arrest), pick and isolate 100 virgin, 1-day-old fog-2 females into a microcentrifuge tube containing 1 mL of M9 buffer. Wash the females 5x with M9 buffer to minimize bacterial contamination and incubate them in 100 µL of M9 buffer for 6 h at 20 °C to allow pheromone production and accumulation in the media.
- Perform a similar extraction process for C. remanei females, using only 15-20 L4 stage females isolated the day before extraction to three plates, and the next day, incubate five virgin females in 100 µL of M9 buffer for 6 h at 25 °C (optimal growth temperature for C. remanei)58.
NOTE: C. remanei females produce a higher amount of volatile sex pheromones compared to C. elegans females. Therefore, only five females are sufficient to extract 100 µL of crude volatile sex pheromone.
- Storage
- Centrifuge samples at 15,000 × g for 30-60 s to pellet the worms. Carefully pipet-transfer the supernatant (containing pheromones) to a clean tube and discard the tube containing the worm pellet. Store the isolated supernatant for subsequent chemoattraction assays.
NOTE: The crude volatile sex pheromone extract can be stored for at least 1 year at -80 °C in a microcentrifuge tube wrapped with parafilm to minimize evaporation.
- Centrifuge samples at 15,000 × g for 30-60 s to pellet the worms. Carefully pipet-transfer the supernatant (containing pheromones) to a clean tube and discard the tube containing the worm pellet. Store the isolated supernatant for subsequent chemoattraction assays.
- Quality control
- Before using the extract, perform a quality control test using N2 males or him-5 males to verify its chemoattractiveness (see section 4).
3. A large amount of crude sex pheromone extraction from 6-day-old virgin hermaphrodites (Figure 1A)
- Worm culture
- To obtain approximately 20 mL of crude C. elegans sex pheromone, synchronize twenty 10 cm NGM plates containing healthy adult C. elegans (either N2 or daf-22 mutant) worms using the aforementioned bleach protocol and then wash them 5x with M9 buffer.
NOTE: This process prepares enough embryos for pheromone extraction. N2: The standard WT C. elegans strain that produces both water-soluble ascaroside pheromones and non-ascaroside volatile sex pheromones. daf-22 mutant: A mutant lacking the ability to produce many ascaroside pheromones, making them helpful in studying non-ascaroside volatile sex pheromones in isolation. - To synchronize worms at the L1 stage, rotate the embryos in M9 buffer for 12-15 h to arrest development. Transfer the arrested L1 worms to 10 cm NGM culture plates seeded with OP50 bacteria for growth and development.
- To minimize the presence of males on hermaphrodite plates, check the plates 2 days after worm release, at the L4 stage, and remove any observed males (very rare).
- After three days of development, look for the appearance of embryos indicating that the worms have become reproductively mature adults.
NOTEfog-2 mutant females of C. elegans and WT females of C. remanei are unsuitable for bulk volatile sex pheromone extraction. fog-2 mutant C. elegans and C. remanei have a high proportion of males in their populations, and males continuously mate with females. Fertilized females do not produce volatile sex pheromones. Male presence complicates the extraction of these compounds.
- To obtain approximately 20 mL of crude C. elegans sex pheromone, synchronize twenty 10 cm NGM plates containing healthy adult C. elegans (either N2 or daf-22 mutant) worms using the aforementioned bleach protocol and then wash them 5x with M9 buffer.
- Worm washing and embryo separation
- Wash the embryos repeatedly with M9 buffer and let them settle (1 g sedimentation) until most of the adults are at the bottom of the tube; repeat 5-7x. Allow the microcentrifuge tubes to settle undisturbed in a rack for several minutes to facilitate the sedimentation of adult worms and enable the separation of the adult worm population from the embryos, which remain suspended in the supernatant.
- Pipette the separated adults and transfer them to new OP50-seeded NGM plates.
- Repeat this washing process for 5-6 days to deplete self-sperm.
NOTE: For the extraction of volatile sex pheromones, utilize hermaphrodites that are 5-6 days old. This timing ensures that their self-sperm is likely to be exhausted, as hermaphrodites with available self-sperm do not produce volatile sex pheromones. Numerous dead or unhatched embryos on the plate by day 5 or 6 indicate an appropriate time to begin pheromone extraction.
- Sex pheromone extraction
- Extract sex pheromone as described in section 1 with modifications.
- Instead of adding 100 C. elegans females per 100 µL, add M9 buffer based on the final worm pellet volume. Add 1 mL of M9 buffer per 50 µL of packed worms.
- Quality control and homogenization
- Quality-control test batches of extracted pheromone using the chemoattraction assay with N2 males or him-5 males (see section 4).
- Mix all batches to create a homogeneous crude volatile sex pheromone for experiments that need a large amount of pheromone, such as screening or microfluidic experiments. Store the crude sex pheromone extract for at least 1 year at -80 °C in 50 mL tubes wrapped with parafilm to minimize evaporation.
- Chemoattraction assay-based titration method for standardization of crude sex pheromone extract
- Perform a titration assay by testing serial dilutions of each pheromone extract on both WT N2 and him-5 male C. elegans. Determine the highest dilution (lowest concentration) that consistently elicits a robust and reproducible chemotaxis response in control strains.
- Prepare a series of dilutions from each crude extract (e.g., 1:2, 1:4, 1:8, etc.) in M9 buffer.
- Chemotaxis assays
- Perform standardized chemotaxis assays (see section 4) using each dilution on the reference strain of males. Perform three replicates to ensure reproducibility. Compare the optimal dilutions across different extraction batches to assess consistency in pheromone concentration.
NOTE: This establishes a reference point for evaluating the bioactivity of each extract. In the standard sex pheromone chemoattraction assay, the original undiluted extracts will be used for subsequent experiments, with the titration serving as a quality control step to ensure consistent pheromone activity between batches. The dilution series can be adjusted based on specific experimental needs. Given that the standard extraction protocol consistently yields saturated crude pheromone extracts, a titration test to standardize the extract may not be necessary for most experiments. It is recommended to use the same batch of crude sex pheromone throughout a set of related experiments to maintain consistency and minimize variability in pheromone activity.
- Perform standardized chemotaxis assays (see section 4) using each dilution on the reference strain of males. Perform three replicates to ensure reproducibility. Compare the optimal dilutions across different extraction batches to assess consistency in pheromone concentration.
4. Volatile sex pheromone chemoattraction assay
NOTE: The volatile sex pheromone chemoattraction assay has been adapted from previously established methods used in other chemoattraction studies24,29,59,60,61. These modifications were implemented to optimize the assay's sensitivity and specificity for detecting responses to volatile sex pheromones. This tailored approach enhances the assay's applicability to specific research needs.
- Observe the culture plates daily until adult female/hermaphrodites are abundant. The health of the worms influences their response to the sex pheromone.
- Preparation of him-5 males
- Utilize the standard bleach protocol to synchronize him-5 worms. After synchronization, wash the worms 5x with M9 buffer. Isolate L4 males the day before the assay; then, transfer one-day-old adult male worms from their seeded plates and rinse them in M9 buffer before the assay. Place them on unseeded NGM plates prior to the assay to eliminate residual bacteria and prevent interference from food during the assay.
NOTE: Do not starve the worms for more than an hour before the assay, as this can alter their internal state and influence the results of the sex pheromone chemoattraction assay. Therefore, if you intend to conduct more than 10 assays in a single day, replace the worm samples every hour. Moreover, alternate between one experimental assay and one control assay repeatedly.
- Utilize the standard bleach protocol to synchronize him-5 worms. After synchronization, wash the worms 5x with M9 buffer. Isolate L4 males the day before the assay; then, transfer one-day-old adult male worms from their seeded plates and rinse them in M9 buffer before the assay. Place them on unseeded NGM plates prior to the assay to eliminate residual bacteria and prevent interference from food during the assay.
- Preparation of chemoattraction agar assay plates
- Prepare chemoattraction assay plates with 1.5% agar, 25 mM NaCl, 1.5 mM Tris-base, and 3.5 mM Tris-Cl as described in relevant literature24.
- Heat the agar in the chemoattraction solution using a microwave until completely dissolved. Allow the solution to cool down at room temperature for 5 min.
- Use a pipette aid to evenly distribute the chemoattraction agar solution into Petri dishes: pour 30 mL into each 10 cm dish and 20 mL into each 6 cm dish.
- After pouring the chemoattraction agar solution into the Petri dishes, leave the lids open in a clean area for at least 40 min to allow the surface of the agar to dry slightly. Once the surface has dried appropriately, close the lids.
NOTE: This drying time may vary depending on the humidity and temperature of the laboratory environment.
- Plate storage and pre-assay preparation
- Box and store the prepared assay plates in a cold room for up to 1 week. Before use, remove the plates from cold storage and allow them to acclimate to room temperature for over an hour. Open the lids to let any residual moisture evaporate from the agar 20 min before the assay in a clean area, ensuring there is no obvious water on the surface before doing the assay.
- Experimental design and chemoattraction assay
- To conduct the chemoattraction assay, mark three distinct spots on the lid and the underside of the Petri dish or use a template printed on transparent plastic paper placed under the dish or attached to the dissecting microscope. These markings include a middle dot (•) as the starting point for the worms, a plus sign (+) for the experimental spot (2 µL of sex pheromone on the lid and 2 µL of 1 M sodium azide on the plate), and a minus sign (-) for the control spot (2 µL of M9 buffer on the lid and 2 µL of 1 M sodium azide) (Figure 2A). Adjust the distance between these marks according to the dish size and specific experimental needs. Typically, for 6 cm Petri dishes, set a fixed distance of 1.5 cm from the starting point to each test substance for the positive control tests.
- Detailed steps to perform the assay (Figure 2)
- Apply 2 µL of 1 M sodium azide to each experimental and control spot on the plate.
- Pick 20 healthy and freely moving male worms with a worm picker. Simultaneously release 20 male worms at the starting point under a dissecting microscope.
- Quickly add 2 µL of sex pheromone and 2 µL of M9 buffer to the experimental and control spots on the lid, respectively.
- Gently close the lid and place the assay plate in a quiet, temperature-stable area next to the microscope.
- After 30 min, score the assay by counting the number of worms at each spot.
NOTE: The process of picking 20 worms should not exceed 1-2 min to prevent the early picked worms from drying out and becoming unhealthy, which could affect the results. The entire process from picking the males to closing the lid should take between 2 and 5 min.
- Positive control screening
- Test the male worms to a 1,000-fold diluted solution of diacetyl (dissolved in 10% ethanol and 90% M9 buffer) to confirm their chemotaxis responsiveness. Evaluate the results of the chemoattraction assay 30 min after initiating the test. Score worms that are paralyzed at the designated spots based on their location: 'C' for those on the control spot, and 'E' for those on the experimental spot. Score worms on neither spot as 'N' (Figure 2C).
- To compute the Chemoattraction Index (C.I.), use the following formula:
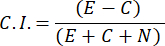
- Select only male samples with a chemotaxis index ≥ 0.4 (C.I., see Figure 2C). Use the male from the same batch for the subsequent sex pheromone test.
NOTE: Conduct three separate assays, each involving 20 worms, to ensure consistency and reliability of the experimental results. Three assays are typically sufficient to infer consistency.
5. Timing and scoring guidelines for chemoattraction assay
- Evaluate the worms' chemoattraction response based on their final locations. Score the number of worms at each spot at the conclusion of the assay, usually 30 min after its beginning.
NOTE: The use of sodium azide at experimental and control spots paralyzes arriving worms, facilitating accurate scoring. Most WT N2 and him-5 males are capable of locating the pheromone source within 6-8 min in a 6 cm plate with a 1.5 cm distance setup. - To capture defects on chemotaxis efficiency, especially to detect subtle defects, consider monitoring the assay at regular intervals of 3-5 min.
NOTE: This frequent observation allows for documentation of the times at which worms arrive at the test spots. Such detailed tracking can reveal cases where worms exhibit decreased chemotaxis efficiency yet still manage to reach the test spot within the examination window.
6. Optional modifications
- For an enhanced assessment of chemotaxis, employ a camera positioned above the plate to record the worms' trajectories throughout the assay.
NOTE: This modification enables a comprehensive analysis of their movement patterns, providing a deeper insight into chemoattraction behavior and trajectory (Figure 3).
7. Data analysis
- Scoring and calculation of Chemoattraction Index (C.I.)
- Follow steps 4.7.1 and 4.7.2.
- Arrival time plot
- For a more comprehensive analysis of chemoattraction assays, consider monitoring the assay at 3-5 min intervals or employing a camera positioned above the plate to record the worms' trajectories throughout the assay.
NOTE: These approaches not only allow scoring the arrival times of the worms at the design spots but also comparing the average arrival times and analyzing the distribution of these times.
- For a more comprehensive analysis of chemoattraction assays, consider monitoring the assay at 3-5 min intervals or employing a camera positioned above the plate to record the worms' trajectories throughout the assay.
- Video-based trajectory analysis and data visualization
NOTE: To analyze the movement patterns of C. elegans during chemoattraction assays, record and extract trajectories (see the Table of Materials) and further analysis (Figure 3).- Set up the recording system.
- Initiate a new recording and access the recording settings window.
- Define the desired recording parameters:
- File Prefix: Choose a descriptive name for the video files (e.g., "experiment1_").
- Frame Rate (FPS): Select the appropriate frames per second for capturing the worm's movement (e.g., 7.5 FPS for worms on the agar plate and 30 FPS for swimming worms).
- Duration: Set the length of the recording in seconds (e.g., 1,800 s [30 min] for bulk chemoattraction assay).
- Optimize the image quality: Adjust the illumination intensity to ensure the worms are clearly visible against the background and fine-tune the focus to acquire a sharp image of the worms.
- To prepare the assay plate, first dispense the control solution onto the designated control spot. Then, carefully pick up the worm(s) and gently release them onto the center of the plate. Once the worm(s) have settled, apply the sex pheromone solution to the designated experimental spot.
- Position the plate: Carefully place the plate containing the worms in the center of the recording field to maximize the capture area.
- To reduce stress on the worms during chemotaxis assays, consider the following two approaches .
- For single worm assay, place the worm on the assay plate and allow it to acclimate for 5 min before introducing the sex pheromone solution at the designated distance (the position based on the worm's current location).
NOTE: This minimizes handling and provides the worm time to adjust to its new environment. - Bulk chemoattraction assay: Allow the worms to settle and disperse naturally on the plate for 5 min. After this acclimation period, introduce the pheromone solution at the designated fixed position. During post video analysis, measure each worm's starting distance from the pheromone and group them accordingly for further analysis.
- For single worm assay, place the worm on the assay plate and allow it to acclimate for 5 min before introducing the sex pheromone solution at the designated distance (the position based on the worm's current location).
- Video analysis procedure
- Import and setup: Import the recorded video into the software. Configure the sequence information (e.g., frame rate); specify the imaging scale (pixels per unit of measurement).
- Image adjustment: To optimize worm detection, adjust the detection threshold until the green label tightly outlines the worms without capturing background noise or artifacts. Additionally, apply background smoothing algorithms to reduce noise and unevenness in the image, enhancing the contrast between the worms and the background. Experiment with different smoothing levels to find the optimal balance between noise reduction and preservation of worm details.
NOTE: The ideal threshold should cover most of each worm's body while excluding extraneous elements. - Detection parameter optimization: Select a representative worm to auto-generate detection parameters. Verify detection accuracy by visually inspecting 5-10 random frames. If detection is unsatisfactory, refine the detection parameters manually, revisit image adjustment settings if needed, and proceed to track once detection is reliable.
- Proofreading and repairing tracking trajectories: After the automated tracking process is complete, perform a manual proofreading of the generated trajectories. If inconsistencies are found during proofreading, use the repair function to correct the trajectory and perform the following operations: Join: Connect trajectory segments that have been incorrectly assigned different identifiers but belong to the same individual. Split: Separate trajectory segments that have been incorrectly assigned the same identifier but belong to different individuals.
NOTE: The goal is to ensure that each unique identifier (number) is consistently assigned to the same individual throughout the entire tracking period.
Example: If a worm is labeled as "3" for a period, then incorrectly labeled as "7" for a subsequent period, the repair function would join these two segments under the identifier "3". Conversely, if two worms are both labeled as "12" for a period, the repair function would split this segment into two separate trajectories, each with a unique identifier. By carefully proofreading and applying necessary repairs, the accuracy and reliability of the tracking data can be significantly improved. - Result visualization and export: Utilize software for visualization and basic analysis. Export data as CSV files for further analysis with the preferred tools or code. This method provides a basic code (see https://github.com/edmondztt/pheromone-traj-analysis.git) to visualize worm movement trajectory based on five key parameters (Figure 3). Time: The progression of the trajectory, color-coded to indicate the passage of time since the start of the experiment. Distance to Pheromone: The distance between the worm and the pheromone source at each time point. Speed: The worm's velocity at each point, which also indicated the turning and stop events. Straightness: How straight the worm's path is. Direction Correctness: How aligned the worm's movement is with the direction of the pheromone target? This code provides a foundational visualization for understanding worm behavior and can be customized and expanded for deeper analysis.
NOTE: This advanced approach provides a comprehensive alternative to the traditional chemoattraction index by detailing worm navigation trajectory in response to sex pheromones. It allows for understanding movement patterns without relying on arbitrary endpoints of the navigation and time window determination, offering deeper insights into the behavioral dynamics of the assay. Data were averaged over 20 frames, which can be adjusted based on specific experimental needs, to filter out movement caused by body twisting.
Results
Trajectory analysis of volatile sex pheromone perception defective strain in chemoattraction assay
This chemoattraction assay reliably differentiates between wild-type and mutant strains of C. elegans in their response to volatile sex pheromones. Successful experiments with him-5 males consistently demonstrate robust chemotaxis towards the pheromone source. This is reflected in a high chemotaxis index (C.I.) (Figure 2), often exceeding 0.5, indicating...
Discussion
This protocol provides a robust methodology for the extraction of volatile sex pheromones from C. elegans, along with establishing a robust chemoattraction assay to measure male chemoattraction responses. Additional information can be found in the WormLab user guide (see the Table of Materials); for a basic code to visualize worm movement trajectory, see protocol section 7.3.8.5. Several crucial steps in the protocol are important for the outcome. First, careful synchronization of worm populatio...
Disclosures
The authors have no conflicts of interest to disclose.
Acknowledgements
We are grateful to Dr. Tingtao Zhou for designing and writing the code for the trajectory visualizations used in our analysis. This work was supported by funding: R01 NS113119 (PWS), Chen senior postdoc fellowship, and the Tianqiao and Chrissy Chen Institute for Neuroscience.
Materials
| Name | Company | Catalog Number | Comments |
| 10 cm Petri dishes | Falcon | 25373-100 | Falcon bacteriological Petri dish 100 x 15 mm |
| 6 cm Petri dishes | Falcon | 25373-085 | Falcon bacteriological Petri dish 60 x 15 mm |
| C. remanei (EM464) | CGC | ||
| Centrifuge | Eppendorf | centrifuge 5418 | Any brand should work. |
| Chemoattraction assay plates | Homemade solution | N/A | 1.5% agar, 25 mM NaCl, 1.5 mM Tris-base, and 3.5 mM Tris-Cl |
| Cholesterol | Alfa Aesar | 57-88-5 | |
| Dissecting Microscope | Leica | LeicaMZ75 | Any brand should work. |
| E. Coli OP50 | CGC | ||
| Ethanol | Koptec | 64-17-5 | |
| fog-2(q71) (JK574) | CGC | ||
| him-5(e1490)(CB4088) | CGC | ||
| Household bleach | Clorox Germicidal bleach concentrated | Bleach | |
| M9 buffer | Homemade solution | N/A | 3 g KH2PO4, 11.3 g Na2HPO4.7H2O, 5 g NaCl, H2O to 1 L. Sterilize by autoclaving. Add 1 mL 1 M MgSO4 after cool down to room temperature. |
| Magnesium Sulfate, Anhydrous, Powder | Macron | M1063-500GM-EA | |
| Microwave | TOSHIBA | N/A | Any brand should work. |
| N2 | CGC | ||
| NaOH | Sigma-aldrich | S318-3 | 1 M |
| NGM plates solution | Homemade solution | N/A | 2.5 g Peptone, 18 g agar, 3 g NaCl, H2O to 1 L.Sterilize by autoclaving. Once the autoclave is done (2 h), wait until the temperature of the medium drops to 65 °C. Put on a hotplate at 65 °C and stir. Then add the following, waiting 5 min between each to avoid crystallization: 1 mL CaCl2 (1 M), 1 mL MgSO4 (1 M), 25 mL K3PO4 (1 M, pH=6), 1 mL Cholesterol ( 5 mg/mL in ethanol). |
| Parafilm | Bemis | 13-374-10 | Bemis Parafilm M Laboratory Wrapping Film |
| Peptone | VWR | 97063-324 | |
| Pipet- aid | Drummond Scientific | 4-000-100 | Any brand should work. |
| Plastic paper | Octago | Waterproof Screen Printing Inkjet Transparency Film | https://www.amazon.com/Octago-Waterproof-Transparency-Printing-Printers/dp/B08HJQWFGD |
| Potassium chloride | Sigma-aldrich | SLBP2366V | |
| Potassium phosphate | Spectrum | 7778-77-0 | |
| Pipette | Eppendorf | SKU: EPPR4331; MFG#: 2231300006 | 20 - 200 µL, 100 - 1000 µL, any brand should work. |
| Rotator | Labnet | SKU: LI-H5500 | Labnet H5500 Mini LabRoller with Dual Direction Rotator. Any brand should work. |
| Sodium chloride | VWR | 7647-14-5 | |
| sodium phosphate dibasic | Sigma-aldrich | SLCG3888 | |
| Tris-base | Sigma-aldrich | 77-86-1 | |
| Tris-Cl | Roche | 1185-53-1 | |
| Tryptone | VWR | 97063-390 | |
| Vortex | Scientific industries | Vortex-Genie 2 | Any brand should work. |
| WormLab system | MBF Bioscience | N/A | https://www.mbfbioscience.com/help/WormLab/Content/home.htm; https://www.mbfbioscience.com/products/wormlab/ |
| Wormpicker | Homemade | N/A | made with platinum and glass pipet tips |
References
- Audesirk, T. E. Chemoreception in Alphysia californica. Iii. Evidence for pheromones influencing reproductive behavior. Behav Biol. 20 (2), 235-243 (1977).
- Traynor, K. S., Le Conte, Y., Page, R. E. Queen and young larval pheromones impact nursing and reproductive physiology of honey bee (Apis mellifera) workers. Behav Ecol Sociobiol. 68 (12), 2059-2073 (2014).
- Cowley, J. J., Wise, D. R. Pheromones, growth and behaviour. Ciba Found Study Group. 35, 144-170 (1970).
- Epple, G. Pheromones in primate reproduction and social behavior. Adv Behav Biol. 11, 131-155 (1974).
- Levinson, H. Z. Possibilities of using insectistatics and pheromones in pest control. Naturwissenschaften. 62 (6), 272-282 (1975).
- Roelofs, W. Manipulating sex pheromones for insect suppression. Environ Lett. 8 (1), 41-59 (1975).
- Marchlewska-Koj, A. Pheromones and mammalian reproduction. Oxf Rev Reprod Biol. 6, 266-302 (1984).
- Keverne, E. B. Neuroendocrinology briefings 11: Pheromones and reproduction. J Neuroendocrinol. 12 (11), 1045-1046 (2000).
- Rekwot, P. I., Ogwu, D., Oyedipe, E. O., Sekoni, V. O. The role of pheromones and biostimulation in animal reproduction. Anim Reprod Sci. 65 (3-4), 157-170 (2001).
- Gomez-Diaz, C., Benton, R. The joy of sex pheromones. EMBO reports. 14 (10), 874-883 (2013).
- Shorey, H. H., Gaston, L. K., Jefferson, R. N. Insect sex pheromones. Adv Pest Control Res. 8, 57-126 (1968).
- Bruce, H. M. Pheromones and behavior in mice. Acta Neurol Psychiatr Belg. 69 (7), 529-538 (1969).
- Shorey, H. H., Bartell, R. J. Role of a volatile female sex pheromone in stimulating male courtship behaviour in Drosophila melanogaster. Anim Behav. 18 (1), 159-164 (1970).
- Bobadoye, B., et al. Evidence of aggregation-sex pheromone use by longhorned beetles (coleoptera: Cerambycidae) species native to Africa. Environ Entomol. 48 (1), 189-192 (2019).
- Saunders, J. R. Sex pheromones in bacteria. Nature. 275 (5682), 692-694 (1978).
- Schulz, S., Toft, S. Identification of a sex pheromone from a spider. Science. 260 (5114), 1635-1637 (1993).
- Finnegan, D. E., Chambers, J. Identification of the sex pheromone of the guernsey carpet beetle, Anthrenussarnicus mroczkowski (coleoptera: Dermestidae). J Chem Ecol. 19 (5), 971-983 (1993).
- Vaillancourt, L. J., Raudaskoski, M., Specht, C. A., Raper, C. A. Multiple genes encoding pheromones and a pheromone receptor define the b beta 1 mating-type specificity in Schizophyllum commune. Genetics. 146 (2), 541-551 (1997).
- Ludewig, A. H., Schroeder, F. C. Ascaroside signaling in C. elegans. WormBook : the online review of C. elegans biology. , 1-22 (2013).
- Sakai, N., et al. A sexually conditioned switch of chemosensory behavior in C. elegans. PLoS One. 8 (7), e68676 (2013).
- Sengupta, S., Smith, D. P., Mucignat-Caretta, C. How Drosophila detect volatile pheromones: signaling, circuits, and behavior. Neurobiology of chemical communication. , (2014).
- Zhang, Y. K., Reilly, D. K., Yu, J., Srinivasan, J., Schroeder, F. C. Photoaffinity probes for nematode pheromone receptor identification. Org Biomol Chem. 18 (1), 36-40 (2019).
- Aprison, E. Z., Ruvinsky, I. Dynamic regulation of adult-specific functions of the nervous system by signaling from the reproductive system. Curr Biol. 29 (23), 4116-4123.e3 (2019).
- Wan, X., et al. SRD-1 in AWA neurons is the receptor for female volatile sex pheromones in C. elegans males. EMBO Rep. 20 (3), e46288 (2019).
- Peckol, E. L., Troemel, E. R., Bargmann, C. I. Sensory experience and sensory activity regulate chemosensory receptor gene expression in Caenorhabditis elegans. Proc Natl Acad Sci U S A. 98 (20), 11032-11038 (2001).
- Jang, H., et al. Neuromodulatory state and sex specify alternative behaviors through antagonistic synaptic pathways in. C. elegans. Neuron. 75 (4), 585-592 (2012).
- Liu, K. S., Sternberg, P. W. Sensory regulation of male mating behavior in Caenorhabditis elegans. Neuron. 14 (1), 79-89 (1995).
- Chasnov, J. R., Chow, K. L. Why are there males in the hermaphroditic species Caenorhabditis elegans. Genetics. 160 (3), 983-994 (2002).
- Chasnov, J. R., So, W. K., Chan, C. M., Chow, K. L. The species, sex, and stage specificity of a Caenorhabditis sex pheromone. Proc Natl Acad Sci U S A. 104 (16), 6730-6735 (2007).
- Susoy, V., et al. Natural sensory context drives diverse brain-wide activity during C. elegans mating. Cell. 184 (20), 5122-5137.e17 (2021).
- Barriere, A., Felix, M. A. High local genetic diversity and low outcrossing rate in Caenorhabditis elegans natural populations. Curr Biol. 15 (13), 1176-1184 (2005).
- Click, A., Savaliya, C. H., Kienle, S., Herrmann, M., Pires-Dasilva, A. Natural variation of outcrossing in the hermaphroditic nematode Pristionchus pacificus. BMC Evol Biol. 9, 75 (2009).
- Anderson, J. L., Morran, L. T., Phillips, P. C. Outcrossing and the maintenance of males within C. elegans populations. J Hered. 101 (suppl_1), S62-S74 (2010).
- Leighton, D. H., Choe, A., Wu, S. Y., Sternberg, P. W. Communication between oocytes and somatic cells regulates volatile pheromone production in Caenorhabditis elegans. Proc Natl Acad Sci U S A. 111 (50), 17905-17910 (2014).
- White, J. Q., et al. The sensory circuitry for sexual attraction in C. elegans males. Curr Biol. 17 (21), 1847-1857 (2007).
- Fagan, K. A., et al. A single-neuron chemosensory switch determines the valence of a sexually dimorphic sensory behavior. Curr Biol. 28 (6), 902-914.e5 (2018).
- Lipton, J., Kleemann, G., Ghosh, R., Lints, R., Emmons, S. W. Mate searching in Caenorhabditis elegans: A genetic model for sex drive in a simple invertebrate. J Neurosci. 24 (34), 7427-7434 (2004).
- Frady, E. P., Palmer, C. R., Kristan, W. B. Sexual attraction: Sex-specific wiring of neural circuitry. Curr Biol. 22 (22), R953-R956 (2012).
- Liu, Q., et al. Two preputial gland-secreted pheromones evoke sexually dimorphic neural pathways in the mouse vomeronasal system. Front Cell Neurosci. 13, 455 (2019).
- Ludewig, A. H., et al. Pheromone sensing regulates Caenorhabditis elegans lifespan and stress resistance via the deacetylase sir-2.1. Proc Natl Acad Sci U S A. 110 (14), 5522-5527 (2013).
- Golden, J. W., Riddle, D. L. A pheromone influences larval development in the nematode Caenorhabditis elegans. Science. 218 (4572), 578-580 (1982).
- Butcher, R. A., Fujita, M., Schroeder, F. C., Clardy, J. Small-molecule pheromones that control dauer development in Caenorhabditis elegans. Nat Chem Biol. 3 (7), 420-422 (2007).
- Jeong, P. Y., et al. Chemical structure and biological activity of the Caenorhabditis elegans dauer-inducing pheromone. Nature. 433 (7025), 541-545 (2005).
- Butcher, R. A., Ragains, J. R., Kim, E., Clardy, J. A potent dauer pheromone component in Caenorhabditis elegans that acts synergistically with other components. Proc Natl Acad Sci U S A. 105 (38), 14288-14292 (2008).
- Aprison, E. Z., Ruvinsky, I. Counteracting ascarosides act through distinct neurons to determine the sexual identity of C. elegans pheromones. Curr Biol. 27 (17), 2589-2599.e3 (2017).
- Aprison, E. Z., Ruvinsky, I. Sex pheromones of C. elegans males prime the female reproductive system and ameliorate the effects of heat stress. PLoS Genet. 11 (12), e1005729 (2015).
- Izrayelit, Y., et al. Targeted metabolomics reveals a male pheromone and sex-specific ascaroside biosynthesis in Caenorhabditis elegans. ACS Chem Biol. 7 (8), 1321-1325 (2012).
- Kaplan, F., et al. Ascaroside expression in Caenorhabditis elegans is strongly dependent on diet and developmental stage. PLoS One. 6 (3), e17804 (2011).
- Pungaliya, C., et al. A shortcut to identifying small molecule signals that regulate behavior and development in Caenorhabditis elegans. Proc Natl Acad Sci U S A. 106 (19), 7708-7713 (2009).
- Srinivasan, J., et al. A modular library of small molecule signals regulates social behaviors in Caenorhabditis elegans. PLoS Biol. 10 (1), e1001237 (2012).
- Srinivasan, J., et al. A blend of small molecules regulates both mating and development in Caenorhabditis elegans. Nature. 454 (7208), 1115-1118 (2008).
- Choe, A., et al. Ascaroside signaling is widely conserved among nematodes. Curr Biol. 22 (9), 772-780 (2012).
- Von Reuss, S. H., et al. Comparative metabolomics reveals biogenesis of ascarosides, a modular library of small-molecule signals in C. elegans. J Am Chem Soc. 134 (3), 1817-1824 (2012).
- Choe, A., et al. Sex-specific mating pheromones in the nematode panagrellus redivivus. Proc Natl Acad Sci U S A. 109 (51), 20949-20954 (2012).
- Hsueh, Y. P., Mahanti, P., Schroeder, F. C., Sternberg, P. W. Nematode-trapping fungi eavesdrop on nematode pheromones. Curr Biol. 23 (1), 83-86 (2013).
- Hong, M., et al. Early pheromone experience modifies a synaptic activity to influence adult pheromone responses of C. elegans. Curr Biol. 27 (20), 3168-3177.e3 (2017).
- Ryu, L., et al. Feeding state regulates pheromone-mediated avoidance behavior via the insulin signaling pathway in Caenorhabditis elegans. EMBO J. 37 (15), e98402 (2018).
- Diaz, S. A., Lindström, J., Haydon, D. T. Basic demography of Caenorhabditis remanei cultured under standard laboratory conditions. J Nematol. 40 (3), 167 (2008).
- Bargmann, C. I. Chemosensation in C. elegans. WormBook : the online review of C.elegans biology. , 1-29 (2006).
- Margie, O., Palmer, C., Chin-Sang, I. C. elegans chemotaxis assay. JoVE: J Vis Exp. (74), e50069 (2013).
- Bargmann, C. I., Hartwieg, E., Horvitz, H. R. Odorant-selective genes and neurons mediate olfaction in C. elegans. Cell. 74 (3), 515-527 (1993).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved