A subscription to JoVE is required to view this content. Sign in or start your free trial.
Metabolomic Analysis of Barley by Gas Chromatography/Mass Spectrometry
In This Article
Summary
A method for metabolomic analysis of barley is presented. The method entails fractionation and derivatization of metabolites and analysis thereof by gas chromatography/mass spectrometry (GC/MS). Metabolomic analysis can be used to determine the effect of intercropping and drought stress on the grain.
Abstract
Climate change increases drought risk to agriculture and impacts both food nutrient content and overall food security. Metabolomics is one way to observe and quantify the impacts of drought on grain and other agricultural products. The identified metabolites may allow for the identification of the biochemical response that allows the plant to tolerate stressful environments. The methodology presented herein allowed for the total metabolomic analysis of barley flour using gas chromatography/mass spectrometry (GC/MS).
Barley flour metabolite extracts were fractionated into four fractions based on polarity. To allow for analysis by GC/MS, metabolites were derivatized to increase volatility and metabolite separation: fatty acids esters were derivatized into fatty acid methyl esters; sugars were oximated into their straight chain form; and metabolites with hydroxyl groups were converted to their corresponding silyl ethers. The derivatized samples were injected into the GC/MS and the generated mass spectra were used for metabolite identification by comparing the generated spectra to the National Institute of Standards and Technology (NIST) Tandem Mass Spectra library. The method described here can also be used to examine the total metabolome for other plants, furthering our understanding of the biochemical responses of stressed plants.
Introduction
Barley is the fourth most produced grain worldwide1. With climate change increasing the frequency and intensity of droughts, the productivity of this crop is expected to drop significantly as severe drought stress can decrease the grain yield by nearly an order of magnitude2. Agronomic practices that diversify the landscape are hypothesized to increase the resilience -- the tendency to maintain yields and soil properties of cultivated land. Intercropping, the cultivation of two crops simultaneously in the same field, is one such practice, especially when nitrogen-fixing pulse crops, like lentils or peas, are included3.
Plant metabolites are small molecules found in plant tissues and cells, such as sugars, amino acids, and lipids, that can be used to understand plant responses to physical and environmental stress. Previous studies have shown certain metabolites in barley, like proline and certain sugars, are present at a higher level in drought conditions, but a full metabolomic study has yet to be done for both drought stress and intercropped conditions4. What remains unclear is how the metabolome of the grain changes due to the biotic stress of intercropping and environmental stress arising from drought. The metabolome -- the total metabolic profile of the grain -- is important for both the end applications of the barley and for identifying the biochemical responses that allow plants to adapt to changes in their environment5.
This study describes the method development and preliminary results of metabolomic analysis of barley grown under drought-stressed and intercropped conditions using gas chromatography/mass spectrometry (GC/MS). For analysis, the barley metabolites were fractionated and derivatized according to polarity into four fractions (Figure 1). The main constituents of the fractions are as follows: Fraction 1: fatty acid methyl esters (FAMEs) and hydrocarbons; Fraction 2: silylated free fatty acids, fatty alcohols, hydroxy FAMEs, phenols, and sterols; Fraction 3: silylated sugars and polyols; and Fraction 4: silylated hydrophilic acids, amino acids, and amines. Thus, Fractions 1 and 2 predominantly contain non-polar metabolites, while Fractions 3 and 4 predominantly contain polar metabolites.
First, metabolites are extracted from the ground barley grain into polar and non-polar solvents. Further fractionation is performed to enable better separation of metabolites in the mass spectra. Additionally, the boiling points of many of the metabolites are too high to allow for volatilization by GC, requiring further derivatization. Thus, metabolites containing hydroxyl groups were silylated into the corresponding trimethylsilyl (TMS) ethers6, while fatty acid esters were converted into fatty acid methyl esters. Additionally, sugars were oximated into their straight-chain form, reducing the number of possible isomers present for each sugar7. Figure 1 shows a flow chart for the fractionation and derivatization of the metabolites in barley flour. The protocol below provides more detail on the specific steps shown in this figure.
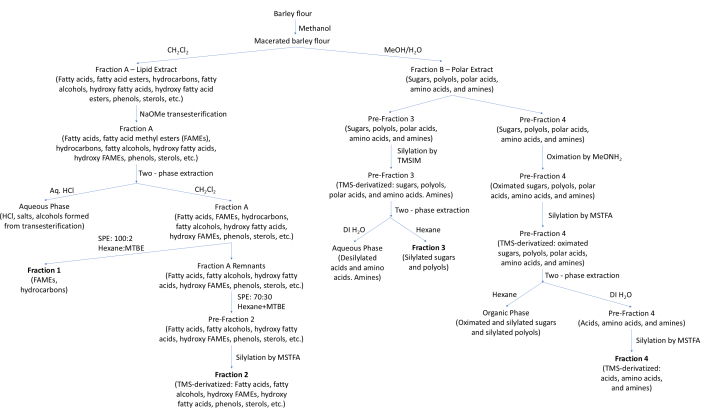
Figure 1: A flow diagram for the fractionation and derivatization of barley metabolites. Abbreviations: MeOH = methanol; NaOMe = sodium methoxide; TMSIM = N-trimethylsilyimidazole; MeONH2 = methoxyamine hydrochloride; MSTFA = N-methyl-N-(trimethylsilyl)trifluoroacetamide; DI H2O = deionized water; MTBE = methyl tert-butyl ether; SPE = solid phase extraction. Please click here to view a larger version of this figure.
Protocol
1. Preparation of standards (Figure 2)
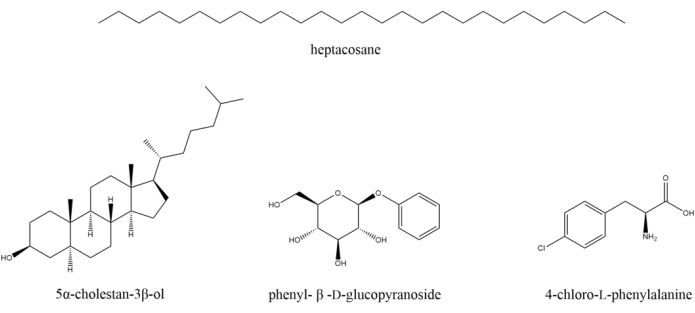
Figure 2: Chemical structures for the four internal standards used in this method. These four standards are used for the semiquantitation of metabolites in the different barley fractions. Please click here to view a larger version of this figure.
- Prepare Standard 1 by dissolving n-heptacosane (50 mg, ≥97% purity) in n-hexane (100 mL, ≥95% purity).
- Prepare Standard 2 by dissolving 5α-cholestan-3β-ol (60 mg, ≥95% purity) in dichloromethane (CH2Cl2, 100 mL, ≥99% purity).
- Prepare Standard 3 by dissolving phenyl-β-D-glucopyranoside (80 mg, ≥95% purity) in deionized water (DI H2O, 100 mL).
- Prepare Standard 4 by dissolving 4-chloro-L-phenylalanine (40 mg, ≥97% purity) in DI H2O (100 mL).
2. Initial fractionation
- Load barley flour (200 mg) into a Solid Phase Extraction (SPE) solvent reservoir. Soak the flour with methanol (200 µL, ACS grade or higher purity) for 20 min to macerate. Remove the methanol through evaporation under vacuum for 30 min (Figure 3).
- Once dried, attach the column to a vacuum manifold. Extract non-polar components (Fraction A) with CH2Cl2 (4 mL) into a 10 mL vial and run the vacuum manifold until the flour is dry.
- Extract the polar components (Fraction B) by adding a mixture of methanol and DI H2O (10 mL, 8:2 v/v). Collect into a 10 mL vial and run the vacuum manifold until the flour is dry.

Figure 3: The vacuum manifold setup. This setup is used to dry the methanol-soaked barley flour. Please click here to view a larger version of this figure.
3. Processing of Fraction A to produce Fractions 1 and 2
- Add standard 1 and standard 2 (100 µL each) to Fraction A and mix by repeated pipetting. Fully evaporate the solvent under reduced pressure using a rotatory evaporator.
- Redissolve the sample in methyl tert-butyl ether (MTBE, 500 µL, ACS grade or higher purity). Add methanol (300 µL) and sodium methoxide (14.6 mg, 97%). Allow transesterification to proceed for 90 min at room temperature under magnetic stirring.
- Add aqueous hydrochloric acid (2 mL, 0.35 M, ACS grade or higher purity). Mix the acidified solution with CH2Cl2 (1 mL). Collect the contents of the vial with a long Pasteur pipette and allow the phases to separate in the pipette.
- Transfer the lower (organic) phase to a new vial. Wash the organic phase by adding aqueous hydrochloric acid (2 mL, 0.35 M). Collect the contents of the vial with a long Pasteur pipette and allow the phases to separate in the pipette.
- Transfer the lower (organic) phase to a new vial. Evaporate the lower (organic) phase to dryness on a rotary evaporator.
- Add anhydrous sodium sulfate (200 mg, ACS grade or higher purity) to an SPE column. Dissolve the sample from step 3.5 in CH2Cl2 (250 µL) and load onto the SPE column.
- Elute Fraction 1 from the SPE column with three portions of n-hexane and MTBE (2.04 mL, 50:1 v/v) into a 10 mL vial. Evaporate the solution containing Fraction 1 (FAMEs and hydrocarbons) to dryness on a rotary evaporator. Redissolve the sample in n-hexane (300 µL), and transfer to an autosampler vial for use on the GC/MS.
- Elute pre-fraction 2 from the SPE column (after elution of Fraction 1 in step 3.7 has been performed) with three subsequent portions of n-hexane and MTBE (2 mL, 7:3 v/v) into a 10 mL vial. Evaporate pre-fraction 2 to dryness on a rotary evaporator.
- Using the same vial, redissolve pre-Fraction 2 in anhydrous pyridine (250 µL, 99.8%) (transfer pyridine using the argon balloon method-see below) and add N-methyl-N-(trimethylsilyl)trifluoroacetamide (MSTFA, 50 µL, Reagent grade).
NOTE: All anhydrous solvents are transferred using the argon balloon method.- Using zip ties, secure a nitrile glove over the end of a syringe with the plunger removed. Fill the balloon with anhydrous argon and attach a disposable needle to the syringe.
- Push the balloon syringe through the septum of the bottle containing the anhydrous reagent and insert a second syringe with a long needle to draw up the solvent (Figure 4).
- Flush the sample vial with a gentle stream of anhydrous argon, cap, seal with parafilm, and place in a 70 °C oil bath under magnetic stirring for 15 min.
- Following the silylation, cool Fraction 2 (TMS-derivatized: fatty acids, fatty alcohols, hydroxy FAMEs, hydroxy fatty acids, phenols, and sterols) at room temperature for 5 min. Load Fraction 2 into an autosampler vial.

Figure 4: The argon balloon setup. Please click here to view a larger version of this figure.
4. Processing of Fraction B to produce Fractions 3 and 4
- Add Standards 3 and 4 (300 µL each) to Fraction B and mix by repeated pipetting. Transfer 2 mL of this solution to a new vial to be processed into Fraction 3 and then transfer 4 mL of this solution to a separate vial to be processed into Fraction 4, starting from the evaporation of pre-fraction 4 in step 4.4. Evaporate pre-fraction 3 to dryness on a rotary evaporator, followed by coevaporation with anhydrous pyridine (2 x 1.5 mL).
NOTE: Coevaporation is used to remove trace amounts of water as anhydrous pyridine forms an azeotrope. - Redissolve pre-fraction 3 in anhydrous pyridine (200 µL) and add N-trimethylsilyimidazole (TMSIM, 100 µL, 97%). Flush the sample vial with a gentle stream of anhydrous argon, cap, seal with parafilm, and heat for 20 min in a 70 °C oil bath under magnetic stirring.
NOTE: TMSIM is a mild silylating agent and will preferentially silylate -OH groups over amine groups8. - Following the silylation, allow the solution to cool at room temperature for 5 min, add n-hexane (200 µL) and DI H2O (400 µL), and stir. Transfer the upper, organic phase, i.e., Fraction 3 (TMS-derivatized sugars and polyols), to an autosampler vial using a long Pasteur pipette.
- Evaporate pre-fraction 4, from step 4.1, to dryness on a rotary evaporator, followed by coevaporation with anhydrous pyridine (2 x 1.5 mL). Redissolve the sample in anhydrous pyridine (250 µL) and add methoxyamine hydrochloride (5 mg, 98%). Flush the sample vial with argon, cap, seal with parafilm, and oximate for 30 min in a 70 °C oil bath under magnetic stirring.
- Allow the solution to cool at room temperature for 5 min, add MSTFA (100 µL), gently flush the sample vial with anhydrous argon, cap, seal with parafilm, and silylate for 20 min in a 70 °C oil bath under magnetic stirring.
- Allow the solution to cool at room temperature for 5 min, then add n-hexane and DI H2O (800 µL, 5:3 v/v). Collect the lower (aqueous) phase using a long Pasteur pipette and wash with 4 x 500 µL of n-hexane, collecting the lower phase and discarding the upper phase every time. Evaporate the aqueous phase to dryness on a rotary evaporator, and coevaporate the remaining material with anhydrous acetonitrile (2 x 1.5 mL, ACS grade or higher purity).
- Redissolve the sample in anhydrous acetonitrile (250 µL) and add MSTFA (50 µL). Gently flush the sample vial with anhydrous argon, cap, seal with parafilm, and heat for 60 min in a 70 °C oil bath under magnetic stirring.
- Allow the solution to cool at room temperature for 5 min, then transfer Fraction 4 (TMS-derivatized carboxylic acids, amino acids, amines) to an autosampler vial.
5. GC/MS methods
- Load samples onto a GC/MS with the following column characteristics: 15 m x 0.25 mm internal diameter, coated with a 0.25 µm nonpolar film.
- Use the following injection volumes for Fractions 1-4, respectively: 1.0, 1.0, 0.5, and 0.5 µL. Run injected samples with a split flow (1:10 split ratio) of 14 mL/min helium at an injection temperature of 250 °C.
- For Fractions 1 and 2, ramp the column temperature from 100 °C to 300 °C at 4 °C/min with a 10 min hold.
NOTE: The total run time is 65 min. - For Fraction 3, heat and hold the column at 300 °C for the entirety of the run.
NOTE: The total run time is 70 min. - For Fraction 4, heat the column from 70 °C to 142 °C at 2.5 °C/min with a 10 min hold, followed by heating from 142 °C to 235 °C at 4 °C/min with a 10 min hold. Finally, bake the column at 320 °C for 8 min.
NOTE: The total run time is 102 min.
6. Metabolite identification
- Perform metabolite identification by comparing each compound's mass spectrum to the NIST mass spectral libraries using chromatography software with search index matching. Select a candidate peak using the computer cursor; then, view the spectra. Right-click on the spectra to initiate the library search.
NOTE: This step can be automated depending on the instrument software. - Semiquantitate each sample by normalizing the peak areas to the standard by dividing the peak area of the sample peak by the peak area of the standard for each fraction.
NOTE: This allows for comparison of metabolite levels across fractions and samples even when sample processing varies.
Results
The developed protocol successfully derivatizes and resolves the many metabolites present in the barley flour. Each peak on the chromatogram is associated with a compound identified from its mass spectra. The three most prominent peaks in Fraction 1 of the sample were octadecadienoic acid methyl ester, hexadecanoic acid methyl ester, and octadecenoic acid. The three most prominent peaks in Fraction 2 of the sample were TMS-derivatized palmitic acid, octadecanoic acid, and n-hexa...
Discussion
We disclose a successful method for the extraction of metabolites from barley grain for metabolic analysis. Several steps in this extraction protocol require particular attention to achieve satisfactory results. In addition, care also needs to be taken during the analysis stage, especially given the number and different properties of the derivatized products.
In order for the metabolite derivatizations to be effective, it is necessary to exclude moisture from solvents and reaction vessels. For...
Disclosures
The authors have no conflicts of interest to declare.
Acknowledgements
Funding was provided by the USDA ARS Pulse Crop Health Initiative and the Idaho Barley Commission.
Materials
| Name | Company | Catalog Number | Comments |
| 4-chloro-L-phenylalanine | Alfa Aesar | 15470329 | 97% |
| 5α-cholestan-3β-ol | Sigma | D6128-10G | 95% |
| Anhydrous acetonitrile | Sigma | 360457 | ACS (Dried over 3Å molecular sieves) |
| Anhydrous Pyridine | Sigma | PX2012 | 99.8% |
| Anhydrous Sodium Sulfate | EMD | SX0760-1 | ACS |
| Argon | |||
| Barley flour | Grown at the Aberdeen Research and Extension Center, University of Idaho | ||
| Deionized Water | |||
| Dichloromethane | TCI | D3478 | 99% |
| Hydrochloric Acid | EMD | HX0603-3 | ACS |
| ISQ 7000 mass spectrometer | ThermoFisher | ||
| Long Pastuer pipet | |||
| Methanol | Macron | 3016-16 | ACS |
| Methoxylamine hydrochloride | Thermo | A19188.06 | 98% |
| Methyl tert-butyl ether | VWR | BDH1139-4LG | ACS |
| n-Heptacosane | Thermo | L07796.MD | 97% |
| n-Hexane | Sigma | HX0295-6 | 95% |
| N-methyl-N-trimethylsilyl-trifluoroacetamide | Restek | 35600 | Reagent |
| N-trimethylsilyimidazole | Alfa Aesar | A12512 | 97% |
| phenyl-β-D-glucopyranoside | Sigma | 292710-5G | 95% |
| Rotary Evaporator | |||
| Sodium Methoxide | Merck | 8.06538.0005 | 97% |
| SPE Column | Agilent | 12113010 | |
| SPE Solvent Resevoir Cartridge | Agilent | 12131014 | |
| TG-SQC column | |||
| Trace 1300 gas chromatograph | ThermoFisher | ||
| Vacuum Manifold |
References
- Verstegen, H., Köneke, O., Korzun, V., von Broock, R., Kumlehn, J., Stein, N. The world importance of barley and challenges to further improvements. Biotechnological approaches to barley improvement. Biotechnology in Agriculture and Forestry. 69, 3-19 (2014).
- . Agribusiness handbook: Barley malt beer Available from: https://www.fao.org/4/i1003e/i1003e00.htm (2009)
- Jena, J., et al. Role of legumes in cropping system for soil ecosystem improvement. Ecosystem services: types, management and benefits. , 1-21 (2022).
- Ahmed, I. M., et al. Differential changes in grain ultrastructure, amylase, protein and amino acid profiles between Tibetan wild and cultivated barleys under drought and salinity alone and combined stress. Food Chem. 141 (3), 2743-2750 (2013).
- Marchev, A. S., et al. Metabolomics and health: from nutritional crops and plant-based pharmaceuticals to profiling of human biofluids. Cell Mol Life Sci. 78 (19-20), 6487-6503 (2021).
- Kusch, P. Gas chromatography: Derivatization, sample preparation, application. IntechOpen. , (2019).
- Haas, M., Lamour, S., Trapp, O. Development of an advanced derivatization protocol for the unambiguous identification of monosaccharides in complex mixtures by gas and liquid chromatography. J Chromatogr A. 1568, 160-167 (2018).
- Moldoveanu, S. C., David, V. Derivatization methods in GC and GC/MS. Gas chromatography-derivatization, sample preparation, application. , (2018).
- Kim, B. R., Sung, G. H., Kim, J. -. J., Yoon, Y. -. J. A development of rapid, practical and selective process for preparation of z-oximes. Journal of the Korean Chemical Society. 57 (2), 295-299 (2013).
- Miyagawa, H., Bamba, T. Comparison of sequential derivatization with concurrent methods for GC/MS-based metabolomics. J Biosci Bioeng. 127 (2), 160-168 (2019).
- Yi, L., et al. MeOx-TMS derivatization for GC-MS metabolic profiling of urine and application in the discrimination between normal C57BL/6J and type 2 diabetic KK-Ay mice. Analytical Methods. 6 (12), 4380-4387 (2014).
- Röhlig, R. M., Eder, J., Engel, K. -. H. Metabolite profiling of maize grain: differentiation due to genetics and environment. Metabolomics. 5, 459-477 (2009).
- Cook, B. I., Mankin, J. S., Anchukaitis, K. J. Climate change and drought: From past to future. Current Climate Change Reports. 4, 164-179 (2018).
- Samarah, N. H. Effects of drought stress on growth and yield of barley. Agronomy for Sustainable Development. 25 (1), 145-149 (2005).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved